Abstract
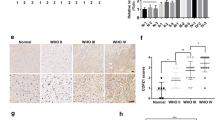
The onconeuronal cerebellar degeneration-related antigen Cdr2 is associated with paraneoplastic syndromes. Neoplastic expression of Cdr2 in ovary and breast tumors triggers an autoimmune response that suppresses tumor growth by developing tumor immunity, but culminates in cerebellar degeneration when Cdr2-specific immune cells recognize neuronal Cdr2. We identified Cdr2 as a novel interactor of the hypoxia-inducible factor (HIF) prolyl-4-hydroxylase PHD1 and provide evidence that Cdr2 might represent a novel important tumor antigen in renal cancer. Strong Cdr2 protein expression was observed in 54.2% of papillary renal cell carcinoma (pRCC) compared with 7.8% of clear-cell RCC and no staining was observed in chromophobe RCC or oncocytoma. High Cdr2 protein levels correlated with attenuated HIF target gene expression in these solid tumors, and Cdr2 overexpression in tumor cell lines reduced HIF-dependent transcriptional regulation. This effect was because of both attenuation of hypoxic protein accumulation and suppression of the transactivation activity of HIF-1α. pRCC is known for its tendency to avascularity, usually associated with a lower pathological stage and higher survival rates. We provide evidence that Cdr2 protein strongly accumulates in pRCC, attenuates the HIF response to tumor hypoxia and may become of diagnostic importance as novel renal tumor marker.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Albert ML, Austin LM, Darnell RB. (2000). Detection and treatment of activated T cells in the cerebrospinal fluid of patients with paraneoplastic cerebellar degeneration. Ann Neurol 47: 9–17.
Albert ML, Darnell RB. (2004). Paraneoplastic neurological degenerations: keys to tumour immunity. Nat Rev Cancer 4: 36–44.
Barth S, Nesper J, Hasgall PA, Wirthner R, Nytko KJ, Edlich F et al. (2007). The peptidyl prolyl cis/trans isomerase FKBP38 determines hypoxia-inducible transcription factor prolyl-4-hydroxylase PHD2 protein stability. Mol Cell Biol 27: 3758–3768.
Berra E, Benizri E, Ginouves A, Volmat V, Roux D, Pouyssegur J. (2003). HIF prolyl-hydroxylase 2 is the key oxygen sensor setting low steady-state levels of HIF-1α in normoxia. EMBO J 22: 4082–4090.
Brown JM, Wilson WR . (2004). Exploiting tumour hypoxia in cancer treatment. Nat Rev Cancer 4: 437–447.
Bruick RK, McKnight SL . (2001). A conserved family of prolyl-4-hydroxylases that modify HIF. Science 294: 1337–1340.
Camenisch G, Tini M, Chilov D, Kvietikova I, Srinivas V, Caro J et al. (1999). General applicability of chicken egg yolk antibodies: the performance of IgY immunoglobulins raised against the hypoxia-inducible factor 1α. FASEB J 13: 81–88.
Coleman ML, McDonough MA, Hewitson KS, Coles C, Mecinovic J, Edelmann M et al. (2007). Asparaginyl hydroxylation of the Notch ankyrin repeat domain by factor inhibiting hypoxia-inducible factor. J Biol Chem 282: 24027–24038.
Corradi JP, Yang C, Darnell JC, Dalmau J, Darnell RB . (1997). A post-transcriptional regulatory mechanism restricts expression of the paraneoplastic cerebellar degeneration antigen cdr2 to immune privileged tissues. J Neurosci 17: 1406–1415.
Cummins EP, Berra E, Comerford KM, Ginouves A, Fitzgerald KT, Seeballuck F et al. (2006). Prolyl hydroxylase-1 negatively regulates IkB kinase-b, giving insight into hypoxia-induced NFkB activity. Proc Natl Acad Sci USA 103: 18154–18159.
Darnell JC, Albert ML, Darnell RB . (2000). Cdr2, a target antigen of naturally occuring human tumor immunity, is widely expressed in gynecological tumors. Cancer Res 60: 2136–2139.
Darnell RB, Posner JB . (2003a). Observing the invisible: successful tumor immunity in humans. Nat Immunol 4: 201.
Darnell RB, Posner JB . (2003b). Paraneoplastic syndromes involving the nervous system. N Engl J Med 349: 1543–1554.
Eble JN, Sauter G, Epstein JI, Sesterhenn IA . (2004). World Health Organization Classification of Tumours: Pathology and Genetics of Tumours of the Male Urinary System and Male Gential Organs. IARC Press, WHO Press, Geneva, Switzerland.
Epstein AC, Gleadle JM, McNeill LA, Hewitson KS, O'Rourke J, Mole DR et al. (2001). C. elegans EGL-9 and mammalian homologs define a family of dioxygenases that regulate HIF by prolyl hydroxylation. Cell 107: 43–54.
Erez N, Milyavsky M, Eilam R, Shats I, Goldfinger N, Rotter V . (2003). Expression of prolyl-hydroxylase-1 (PHD1/EGLN2) suppresses hypoxia inducible factor-1α activation and inhibits tumor growth. Cancer Res 63: 8777–8783.
Ferguson 3rd JE, Wu Y, Smith K, Charles P, Powers K, Wang H et al. (2007). ASB4 is a hydroxylation substrate of FIH and promotes vascular differentiation via an oxygen-dependent mechanism. Mol Cell Biol 27: 6407–6419.
Ivan M, Haberberger T, Gervasi DC, Michelson KS, Gunzler V, Kondo K et al. (2002). Biochemical purification and pharmacological inhibition of a mammalian prolyl hydroxylase acting on hypoxia-inducible factor. Proc Natl Acad Sci USA 99: 13459–13464.
Jaakkola P, Mole DR, Tian YM, Wilson MI, Gielbert J, Gaskell SJ et al. (2001). Targeting of HIF-α to the von Hippel–Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science 292: 468–472.
Jiang BH, Zheng JZ, Leung SW, Roe R, Semenza GL . (1997). Transactivation and inhibitory domains of hypoxia-inducible factor 1α. Modulation of transcriptional activity by oxygen tension. J Biol Chem 272: 19253–19260.
Koditz J, Nesper J, Wottawa M, Stiehl DP, Camenisch G, Franke C et al. (2007). Oxygen-dependent ATF-4 stability is mediated by the PHD3 oxygen sensor. Blood 110: 3610–3617.
Kononen J, Bubendorf L, Kallioniemi A, Barlund M, Schraml P, Leighton S et al. (1998). Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat Med 4: 844–847.
Kosary CL, McLaughlin JK . (1993). Kidney and renal pelvis. In: Miller BA, Ries LAG, Hankey BF (eds). SEER Cancer Statistics Review, 1973–1990. National Cancer Institute, Bethesda, MD, USA (NIH publication no. 93-2789, XI.1-XI.22).
Kovacs G, Akhtar M, Beckwith BJ, Bugert P, Cooper CS, Delahunt B et al. (1997). The Heidelberg classification of renal cell tumours. J Pathol 183: 131–133.
Mahon PC, Hirota K, Semenza GL . (2001). FIH-1: a novel protein that interacts with HIF-1α and VHL to mediate repression of HIF-1 transcriptional activity. Genes Dev 15: 2675–2686.
Martin F, Linden T, Katschinski DM, Oehme F, Flamme I, Mukhopadhyay CK et al. (2005). Copper-dependent activation of hypoxia-inducible factor (HIF)-1: implications for ceruloplasmin regulation. Blood 105: 4613–4619.
Maxwell PH, Wiesener MS, Chang GW, Clifford SC, Vaux EC, Cockman ME et al. (1999). The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 399: 271–275.
Moch H, Mihatsch MJ . (2002). Genetic progression of renal cell carcinoma. Virchows Arch 441: 320–327.
Motzer RJ, Bander NH, Nanus DM . (1996). Renal-cell carcinoma. N Engl J Med 335: 865–875.
Oliver RT, Nethersell AB, Bottomley JM. (1989). Unexplained spontaneous regression and alpha-interferon as treatment for metastatic renal carcinoma. Br J Urol 63: 128–131.
Onishi T, Oishi Y, Goto H, Yanada S, Abe K . (2002). Histological features of hypovascular or avascular renal cell carcinoma: the experience at four university hospitals. Int J Clin Oncol 7: 159–164.
Ozer A, Wu LC, Bruick RK . (2005). The candidate tumor suppressor ING4 represses activation of the hypoxia inducible factor (HIF). Proc Natl Acad Sci USA 102: 7481–7486.
Peterson K, Rosenblum MK, Kotanides H, Posner JB . (1992). Paraneoplastic cerebellar degeneration. I. A clinical analysis of 55 anti-Yo antibody-positive patients. Neurology 42: 1931–1937.
Pouysségur J, Dayan F, Mazure NM . (2006). Hypoxia signalling in cancer and approaches to enforce tumour regression. Nature 441: 437–443.
Pugh CW, O'Rourke JF, Nagao M, Gleadle JM, Ratcliffe PJ . (1997). Activation of hypoxia-inducible factor-1; definition of regulatory domains within the a subunit. J Biol Chem 272: 11205–11214.
Rolfs A, Kvietikova I, Gassmann M, Wenger RH . (1997). Oxygen-regulated transferrin expression is mediated by hypoxia-inducible factor-1. J Biol Chem 272: 20055–20062.
Santomasso BD, Roberts WK, Thomas A, Williams T, Blachere NE, Dudley ME et al. (2007). A T cell receptor associated with naturally occurring human tumor immunity. Proc Natl Acad Sci USA 104: 19073–19078.
Schofield CJ, Ratcliffe PJ. (2004). Oxygen sensing by HIF hydroxylases. Nat Rev Mol Cell Biol 5: 343–354.
Shin DH, Li SH, Chun YS, Haung LE, Kim MS, Park JW . (2008). CITED2 mediates the paradoxical responses of HIF-1α to proteasome inhibition. Oncogene 27: 1939–1944.
Stiehl DP, Wirthner R, Koditz J, Spielmann P, Camenisch G, Wenger RH . (2006). Increased prolyl 4-hydroxylase domain proteins compensate for decreased oxygen levels. Evidence for an autoregulatory oxygen-sensing system. J Biol Chem 281: 23482–23491.
Struckmann K, Mertz K, Steu S, Storz M, Staller P, Krek W et al. (2008). pVHL co-ordinately regulates CXCR4/CXCL12 and MMP2/MMP9 expression in human clear-cell renal cell carcinoma. J Pathol 214: 464–471.
Takeda K, Ho VC, Takeda H, Duan LJ, Nagy A, Fong GH. (2006). Placental but not heart defects are associated with elevated hypoxia-inducible factor α levels in mice lacking prolyl hydroxylase domain protein 2. Mol Cell Biol 26: 8336–8346.
To KK, Huang LE. (2005). Suppression of hypoxia-inducible factor 1α (HIF-1α) transcriptional activity by the HIF prolyl hydroxylase EGLN1. J Biol Chem 280: 38102–38107.
Vogelzang NJ, Priest ER, Borden L. (1992). Spontaneous regression of histologically proved pulmonary metastases from renal cell carcinoma: a case with 5-year followup. J Urol 148: 1247–1248.
Wanner RM, Spielmann P, Stroka DM, Camenisch G, Camenisch I, Scheid A et al. (2000). Epolones induce erythropoietin expression via hypoxia-inducible factor-1α activation. Blood 96: 1558–1565.
Wenger RH . (2002). Cellular adaptation to hypoxia: O2-sensing protein hydroxylases, hypoxia-inducible transcription factors, and O2-regulated gene expression. FASEB J 16: 1151–1162.
Wenger RH, Stiehl DP, Camenisch G . (2005). Integration of oxygen signaling at the consensus HRE. Sci STKE 306: re12.
Acknowledgements
We would like to thank R Fischer and C Rüber (ETH Zürich, Switzerland) for the generation of monoclonal anti-Cdr2 antibodies, DM Katschinski (Georg August University Göttingen, Germany) for providing the custom-made mouse testis cDNA library, J Nesper (University of Konstanz, Germany) and MO Hottiger (University of Zürich, Switzerland) for help with the yeast two-hybrid screenings, DJ Peet (University of Adelaide, Australia) and N Sang (Thomas Jefferson University, PA, USA) for gifts of plasmids, P Carmeliet and M Schneider (Katholieke Universiteit Leuven and Flanders Institute for Biotechnology, Belgium) for Phd1−/− MEFs, I Flamme and F Oehme (Bayer HealthCare, Germany) for helpful discussions and P Spielmann, M Storz and S Behnke for excellent technical assistance. This work was supported by SNF 3100AO-116047/1 (RHW and GC), Sassella Stiftung (GC, KB and SB), Krebsliga des Kantons Zürich (GC), UZH University Research Priority Program ‘Integrative Human Physiology’ and the 6th Framework Programme of the European Commission EUROXY LSHCCT-2003-502932/SBF 03.0647-2 (RHW).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Balamurugan, K., Luu, VD., Kaufmann, M. et al. Onconeuronal cerebellar degeneration-related antigen, Cdr2, is strongly expressed in papillary renal cell carcinoma and leads to attenuated hypoxic response. Oncogene 28, 3274–3285 (2009). https://doi.org/10.1038/onc.2009.186
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.186
Keywords
This article is cited by
-
MicroRNA 452 regulates ASB8, NOL8, and CDR2 expression in colorectal cancer cells
Genes & Genomics (2021)
-
Cerebellar degeneration-related proteins 2 and 2-like are present in ovarian cancer in patients with and without Yo antibodies
Cancer Immunology, Immunotherapy (2017)
-
Suppression of Calbindin-D28k Expression Exacerbates SCA1 Phenotype in a Disease Mouse Model
The Cerebellum (2012)
-
Differential vascular expression and regulation of oncofetal tenascin-C and fibronectin variants in renal cell carcinoma (RCC): implications for an individualized angiogenesis-related targeted drug delivery
Histochemistry and Cell Biology (2012)
-
Prolyl-4-hydroxylase PHD2- and hypoxia-inducible factor 2-dependent regulation of amphiregulin contributes to breast tumorigenesis
Oncogene (2011)