Article Text
Abstract
Objective To assess feasibility of overnight closed-loop therapy in young children with type 1 diabetes and contrast closed loop using diluted versus standard insulin strength.
Research design and methods Eleven children (male 6; age range 3.75–6.96 years; glycated hemoglobin 60 (14) mmol/mol; body mass index SD score 1.0 (0.8); diabetes duration 2.2 (1.0) years, mean (SD); total daily dose 12.9 (10.6, 16.5) IU/day, median (IQR)) were studied at a clinical research facility on two occasions. In random order, participants received closed loop with diluted insulin aspart (CL_Dil; 20 IU/mL) or closed loop with standard aspart (CL_Std; 100 IU/mL) from 17:00 until 8:00 the following morning. Children consumed an evening meal at 17:00 (44 (12) gCHO) and an optional bedtime snack (6 (7) gCHO) identical on both occasions. Meal insulin boluses were calculated by standard pump bolus calculators. Basal rates on insulin pump were adjusted every 15 min as directed by a model-predictive-control algorithm informed by a real-time glucose sensor values.
Results Mean plasma glucose was 122 (24) mg/dL during CL_Dil vs 122 (23) mg/dL during CL_Std (p=0.993). The time spent in the target glucose range 70–145 mg/dL was 83 (70, 100)% vs 72 (54, 81)% (p=0.328). Time above 145 mg/dL was 13 (0, 27)% vs 19 (10, 45)% (p=0.477) and time spent below 70 mg/dL was 0.0 (0.0, 1.4)% vs 1.4 (0.0, 11.6)% (p=0.161). One asymptomatic hypoglycemia below 63 mg/dL occurred in one participant during CL_Dil versus six episodes in five participants during CL_Std (p=0.09). Glucose variability measured by CV of plasma glucose tended to be reduced during CL_Dil (20% (13, 31) vs 32% (24, 42), p=0.075).
Conclusions In this feasibility study, closed-loop therapy maintained good overnight glucose control with tendency towards reduced hypoglycemia and reduced glucose variability using diluted insulin.
Trial registration number clinicaltrials.gov Identifier: NCT01557634.
- Artificial Pancreas
- Type 1
- Child
This is an Open Access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
Statistics from Altmetric.com
Key messages
-
Closed-loop insulin delivery is a promising medical innovation for treatment of type 1 diabetes.
-
Closed loop may be particularly beneficial for preschool children, who are more vulnerable to glycemic fluctuations and are at greater risk of hypoglycemia.
-
Safe and efficacious overnight glucose control was attained during closed loop using diluted and standard insulin strength, with tendency to reduced hypoglycemia and glycemic variability with diluted insulin.
Closed-loop insulin delivery is an emerging medical innovation which aims to improve glycemic control while reducing the risk of hypoglycemia in type 1 diabetes.1 Closed-loop systems combine glucose-sensing and insulin-delivery components to modulate delivery of insulin in a glucose-responsive fashion. The efficacy and safety of closed-loop therapy has been demonstrated in clinical studies performed in adolescents and children older than 6 years.2–5 To date, one study evaluated closed-loop therapy in children younger than 7 years, using a proportional-integral-derivative control approach.6
Further closed-loop research in younger children is warranted. The incidence of type 1 diabetes is increasing particularly in this age group,7–9 younger children are more prone to hypoglycemic seizures especially overnight,10 and the developing brain appears to be more susceptible to the adverse effects of severe hypoglycemia.11 A longer life-time exposure to hyperglycemia may lead to an accentuated risk of late microvascular and macrovascular complications. An early onset of diabetes has also been more strongly associated with impaired cognitive function12–15 and reduced school achievements,16 with recent evidence of dysglycemia-related anatomical brain changes in young children with type 1 diabetes.17 ,18 Fear of hypoglycemia is common19 and is associated with increased psychological burden and reduced quality of life for children and their families,20 leading to higher than optimal glucose levels as an attempt to prevent hypoglycemia.21 ,22 Glycemic control in preschool children is often suboptimal, even when applying continuous glucose monitoring combined with insulin pump or multiple daily injections.23 ,24 High insulin sensitivity as well as unpredictable food intake and physical activity25 complicate insulin dose adjustments. Infants and young children require very small doses of insulin that challenge the accuracy of subcutaneous insulin administration with standard concentration of 100 IU/mL.26
The purpose of our feasibility study was to evaluate overnight closed-loop therapy with model predictive control in young children with type 1 diabetes and to compare closed loop using diluted versus standard insulin strength. We hypothesized that diluted insulin may lead to more stable glucose levels by reducing inaccuracies in insulin delivery accentuated during the delivery of small amounts of insulin. These inaccuracies result from electromechanical issues such as pump plunger microjumps, insulin reservoir manufacturing tolerances, in-line infusion set pressure variations, and infusion set kinking.
Research design and methods
An open label randomized two-period crossover study compared overnight closed-loop insulin delivery using diluted versus standard insulin strength. The study was approved by the East of England—Cambridge Central Research Ethics Committee. Participants provided assent to the study procedures and informed consent was signed by a parent/carer.
Subjects and study protocol
The study was conducted at the Wellcome Trust Clinical Research Facility at Addenbrooke's Hospital, Cambridge, between December 2012 and November 2013. Children aged 3–6 years were recruited from three pediatric diabetes clinics at Cambridge, London University College Hospital, and Peterborough. Eligibility criteria included type 1 diabetes (WHO criteria) for at least 6 months, insulin pump therapy with good knowledge of insulin dose adjustment by carers, and glycated hemoglobin (HbA1c) below 108 mmol/mol (12%). Exclusion criteria were any physical or psychological disease likely to interfere with the normal conduct of the study and data interpretation or current treatment with drugs likely to interfere with glucose metabolism.
Participants attended the clinical research facility for two overnight periods, 2–6 weeks apart, with identical study protocol performed on both occasions. On one occasion, participants underwent closed-loop therapy using diluted insulin (20 IU/mL) and on the other occasion closed loop with standard insulin strength (100 IU/mL). The order of the interventions was random according to a computer-generated allocation sequence with permuted blocks placed in sealed envelopes.
Continuous glucose monitoring was started 24–72 h before each study visit by inserting a single glucose sensor (Dexcom G4, Dexcom Inc, California, USA) into the subcutaneous tissue of the upper buttocks. Calibration followed manufacturer's instructions using finger-stick glucose measurements taken every 12 h on CONTOUR XT Meter (Bayer, Leverkusen, Germany) which was checked for accuracy by calibration fluid.
On each occasion, participants were admitted at 15:30 and stayed until 8:00 the following day. On admission, participant's insulin pump was replaced by a study pump (Animas 2020, Johnson & Johnson, Pennsylvania, USA) connected to the existing infusion site. A new subcutaneous catheter was inserted 12–24 h before each study visit. An intravenous cannula was placed for blood sampling starting at 16:30. Participants consumed an evening meal at 17:00 (44 (12) g carbohydrates) and an optional bedtime snack (6 (7) g carbohydrates). The meals were identical on the two occasions. Meals and carbohydrate content were chosen by the children and their families based on individual preferences and reflecting usual practice at home. Meals were accompanied by insulin boluses calculated using participants’ standard insulin pump bolus calculator settings and premeal finger-stick glucose levels.
Overnight sensor glucose levels were also recorded at home before each study visit while children maintained their usual exercise and eating patterns and standard insulin pump settings were applied.
Closed loop and insulin dilution
An algorithm based on model predictive control was used to adjust basal insulin delivery during both study visits.27 The operation of closed-loop therapy included manual transfer of sensor glucose data and adjustment of pump basal insulin infusion as advised by the computer-based algorithm at 15 min intervals, as described previously.5 This continued from 17:00 until 8:00 the next day for 15 h.
The algorithm was initialized using participant's weight, total daily insulin dose, and basal insulin infusion. Additionally, the algorithm was provided with sensor glucose levels measured during a 30 min period preceding the start of closed-loop therapy, the carbohydrate content of meals, and prandial insulin boluses. No plasma glucose data were provided to the algorithm. Further details of the closed-loop algorithm are provided elsewhere;5 we used algorithm V.0.03.26.
During closed loop, rapid acting insulin analog aspart (Novo Nordisk, Bagsvaerd, Denmark) was infused either at standard strength (100 IU/mL) or diluted by 0.9% saline at 1:5 ratio resulting in an insulin concentration of 20 IU/mL.
Sampling and assays
Venous blood samples were obtained every 30–60 min for the measurement of glucose and insulin concentration. Plasma was separated by centrifugation immediately. Plasma glucose levels were determined in real time by YSI2300 STAT Plus analyzer (Yellow Springs Instrument, Farnborough, UK) but were not used to inform the algorithm. Plasma insulin concentration was measured by immunochemiluminometric assay (Invitron, Monmouth, UK; intra-assay coefficient of variation (CV), 4.7%; interassay CV, 7.2–8.1%).
Statistical analysis
The primary outcome was the time with plasma glucose levels within the target range from 70 to 145 mg/dL in the overnight period from 21:00 until 8:00 on the following day. Secondary outcomes included time when glucose concentration was below 70 mg/dL (hypoglycemia) and glucose variability as measured by the SD of glucose, the CV of glucose, and mean amplitude of glucose excursions (MAGE).28 Safety measures included hypoglycemia events (plasma glucose below 63 mg/dL) and the low blood glucose index.29 Overnight sensor glucose levels collected at home before study visits were compared with sensor glucose levels measured during closed-loop insulin delivery between 21:00 and 8:00.
Outcomes were calculated using time-weighted data by GStat software, V.2.0 (University of Cambridge, UK). Statistical analyses were conducted using SPSS V.21 (IBM SPSS, Chicago, Illinois, USA). Wilcoxon signed-rank test was used to compare non-normally distributed indices and paired t test to compare normally distributed data. Results are presented as median (IQR) or mean (SD) unless stated otherwise. p values less than 0.05 were considered statistically significant.
Results
Participants
Fourteen participants were consented and randomized. Three participants withdrew after randomization, one at the start of the first study visit and two at the start of the second visit, due to failed attempts to establish intravenous sampling cannula. The study flow chart is shown in online supplementary figure S1. Eleven participants completed the study ((male 6; age 5.07 (1.12), range 3.75–6.96 years; HbA1c 60 (14) mmol/mol; body mass index SD score 1.0 (0.8), range −0.55, 2.11; duration of diabetes 2.2 (1.0) years; total daily dose 12.9 (10.6, 16.5) IU/day and 0.65 (0.59, 0.69) IU/kg/day).
Overnight glucose control
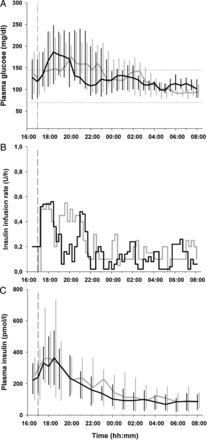
Study outcomes during the overnight period are summarized in table 1. Plasma glucose, insulin delivery and plasma insulin during closed loop with standard insulin strength and closed loop with diluted insulin are shown in figure 1.
Study outcomes
Plasma glucose (A), insulin infusion rates (B), and plasma insulin (C) are shown for closed loop with standard insulin strength (grey line) and closed loop with diluted insulin (black line; median (IQR)). Horizontal dashed lines illustrate the target glucose levels between 70 and 145 mg/dL. The vertical dashed line indicates when closed loop started and the evening meal was consumed.
Plasma glucose levels remained within the target range of 70–145 mg/dL for 72% and 83% of the time, respectively, when closed loop with standard insulin strength and closed loop with diluted insulin was applied (p=0.328; table 1). No difference was found in the mean plasma glucose concentration (122 (23) vs 122 (24) mg/dL, p=0.993). A trend towards reduced time spent in hypoglycemia was observed during closed loop with diluted insulin (plasma glucose less than 70 mg/dL, p=0.161). A reduced low blood glucose index supported this observation (p=0.050). Sensor-based assessment indicated significant reduction of time below 63 mg/dL (p=0.028) and a reduced low blood glucose index (p=0.026; see online supplementary table S1).
Insulin infusion rates were not different during the two interventions (0.2 (0.2, 0.3) vs 0.2 (0.1, 0.3) U/h, p=0.328) and resulted in similar plasma insulin levels (p=0.213).
Hypoglycemia and glucose variability
No episode of hypoglycemia requiring treatment occurred during either intervention. One asymptomatic episode of hypoglycemia below 63 mg/dL as measured by plasma glucose occurred in one participant during closed loop with diluted insulin as compared with six episodes in five participants during closed loop with standard insulin (p=0.09). All these episodes resolved with no carbohydrate intake within 15–45 min.
All measurements of glycemic variability showed a tendency towards reduced variability during closed loop with diluted insulin, although these differences were not statistically significant (table 1).
Sensor accuracy
The median relative absolute difference of Dexcom G4 continuous glucose monitor was 11.6% (6.6, 18.8). The Clarke error grid analysis30 (see online supplementary figure S2) provided 98.6% of the values were in zone A+B, 0% in zone C, 1.4% in zone D, and 0% in zone E.
Glycemic control at home
Comparison between sensor glucose during conventional insulin pump therapy at home settings and closed-loop therapy using standard and diluted insulin is summarized in table 2.
Comparison between glucose control based on sensor glucose during standard insulin pump therapy at home the night before the study visits and during closed loop with diluted and standard insulin at the clinical research facility
Conclusions
We show that closed-loop therapy using a model predictive control approach can be safely and effectively applied for overnight glucose control in young children aged 3–6 years with type 1 diabetes. Plasma glucose levels were maintained between 70 and 145 mg/dL for 72% and 83% of the time using closed loop with standard and diluted insulin, respectively, and mean glucose was 122 mg/dL during both interventions. No episodes of hypoglycemia requiring treatment occurred over 22 study nights.
The present study builds on previous observations in older children and adolescents about benefits of overnight closed-loop therapy using the model predictive control at the clinical research facility5 and in the unsupervised home settings.31 In the present study, we document good glucose control in younger children, who may greatly benefit from closed-loop therapy. Similar results in terms of proportion of time spent in the target range and in hypoglycemia were obtained in this age group as compared with older children and adolescents.32 ,33 The daily management of diabetes in a young child is complicated by amplified responsiveness to insulin, increased risk of nocturnal hypoglycemia, high glucose variability and unpredictability of food intake and physical activity. While the avoidance of hypoglycemia is a crucial concern, chronic hyperglycemia and glucose variability have been suggested to be equally detrimental for brain development and may affect cognition.18 Children younger than 4 years were found to spend more than 4 h/day with glucose levels greater than 250 mg/dL when continuous glucose monitoring was applied.25 Similarly, no evidence of improved glycemic control with insulin pump therapy has been demonstrated in younger children34 possibly owing to the small number of studies targeting this age group.
Closed-loop insulin delivery using a proportional-integral-derivative approach in children younger than 7 years was investigated by Dauber et al6 documenting reduced overnight exposure to hyperglycemia as compared with conventional insulin pump therapy. Although closed loop was not associated with improvements in the time spent in target or with reduced frequency of hypoglycemia, the study was the first to evaluate closed-loop therapy in younger children.
The novelty of our study is the use of diluted insulin to enhance the accuracy of delivery of small insulin doses. When compared with standard insulin strength, diluted insulin during closed-loop therapy tended to reduce the risk of hypoglycemia as evaluated by the low blood glucose index (p=0.050) and time spent in hypoglycemia less than 70 mg/dL (p=0.161). Sensor-based assessment of hypoglycemia showed a significant reduction in the time spent in hypoglycemia below 63 mg/dL (p=0.028) and the low blood glucose index (p=0.026) suggesting that sensor errors attenuated the differences when outcomes were measured by reference plasma glucose.35 Sensor accuracy data are sparse in this young age group. Episodes of asymptomatic self-resolving mild hypoglycemia tended to be lower during closed loop using diluted insulin (p=0.09).
We observed a tendency towards reduced glycemic variability during closed loop with diluted insulin. The median CV of glucose was 20% during closed loop with diluted insulin versus 32% when standard strength insulin was used (p=0.075). Other measures of glucose variability confirmed this trend (table 1). These CVs are lower than those reported by Tsalikian et al,23 who observed a median CV of glucose between 35% and 45% in children younger than 4 years using continuous glucose monitoring, and by Slover et al36 during sensor-augmented pump therapy. Glycemic variability has been inconsistently associated with the risk of vascular complications in diabetes.37 In younger children, increased glycemic variability along with chronic exposure to hyperglycemia have been recently found to be associated with changes in the brain's white matter.17
We chose a dilution so that the volume of insulin scales from 10 U/day of standard strength insulin to 50 U/day of diluted insulin, a common amount applicable in adults. The latter converts to basal insulin delivery of about 1 U/h, a rate at which accuracy of insulin pump is assessed and optimized. A greater dilution may further increase accuracy by limiting the effect of microbubbles and to overcome tissue pressure build-up but greater dilutions may be an issue in children requiring a larger amount of insulin due to pain at the cannula site.
The strength of the study is the randomized study design. The lack of control therapy is a limitation motivated by practical reasons to reduce burden for young participants and taking into account existing evidence of suboptimal glycemic control with frequent hyperglycemic excursions23 ,24 and undetected hypoglycemia38 in younger children on standard therapy. We carried out a post hoc analysis and although detailed information on children's schedule at home, that is, the time of evening meal/bedtime the night before the study was not available, our data showed that glucose control during conventional pump therapy in the home settings appeared inferior to that obtained during closed-loop therapy using standard as well as diluted insulin (table 2).
In conclusion, the present exploratory study suggests that closed-loop insulin delivery using the model predictive control approach is safe and efficacious to maintain overnight glycemic control in young children with type 1 diabetes. Closed-loop therapy using insulin dilution tended to reduce hypoglycemia and glycemic variability as compared to closed loop with standard insulin strength. The use of insulin dilution to enhance accuracy of insulin delivery may be explored in larger studies. Difficulties related to the use of insulin dilution, that is, the need to prepare diluted insulin every 7–14 days and the availability of specific software on insulin pumps to avoid dosing errors, may also need to be taken into account. Our findings support research into closed-loop therapy in preschool children, who may greatly benefit from this novel therapeutic approach.
Acknowledgments
The authors are grateful to the study volunteers for their participation and to staff at the Wellcome Trust Clinical Research Facility for their help in conducting the studies. Dr Vijith Puthi (Peterborough Hospital) and Professor Peter Hindmarsh (University College, London) helped identifying potential recruits. Animas supplied study pumps. Josephine Hayes (Institute of Metabolic Science, University of Cambridge) provided administrative support. Karen Whitehead (Department of Paediatrics, University of Cambridge) provided laboratory support. The Diabetes Research Network Laboratory Wales (Dr Steve Luzio) measured plasma insulin.
References
Supplementary materials
Supplementary Data
This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
Files in this Data Supplement:
- Data supplement 1 - Online supplement
Footnotes
-
Contributors DE, MT, and RH had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. RH coordinated the study. DE, RH, DBD, CLA, and JMA co-designed the studies. DE, JMA, RE-K, PB-A, and MT were responsible for screening and enrolment of participants and arranged informed consent from the participants, provided patient care, collected the clinical and laboratory data, and contributed to biochemical analysis. DE, MT, and RH carried out or supported the data analysis, including the statistical analyses. RH designed and implemented the glucose controller. RH, DBD, DE, MT, and CLA contributed to the interpretation of the results and the writing and critical review of the report.
-
Funding This work was funded by the Juvenile Diabetes Research Foundation (JDRF Grant Number: 22–2011–668) and supported by NIHR Cambridge Biomedical Research Centre.
-
Competing interests RH reports having received speaker honoraria from Minimed Medtronic, Lifescan, Eli Lilly, and Novo Nordisk, serving on advisory panel for Animas and Minimed Medtronic, receiving license fees from BBraun and Beckton Dickinson; and having served as a consultant to Beckton Dickinson, BBraun and Profil. RH and DBD report patent applications.
-
Patient consent Obtained.
-
Ethics approval East of England—Cambridge Central Research Ethics Committee.
-
Provenance and peer review Not commissioned; externally peer reviewed.