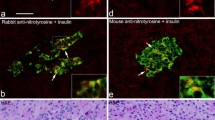
Summary
Autoantibodies reacting with human pancreatic exocrine cells were investigated by immunofluorescent techniques in 107 patients with Type 1 (insulin-dependent) diabetes mellitus, 20 first-degree relatives of the Type 1 diabetic patients, 347 patients with Type 2 (non-insulin-dependent) diabetes, 34 with alcoholic pancreatitis, 26 with rheumatoid arthritis and 107 normal control subjects. Both immunoblotting analysis and double-immunostaining methods were used to characterize the antigens targeted by the pancreatic exocrine cell autoantibodies. Sera positive for human pancreatic exocrine cell cytoplasm, producing a “fine fibrillar” pattern, were found in 21% (23/107) of the Type 1 diabetic patients. The autoantibodies were present in 39% (15/38) of Type 1 diabetic patients diagnosed within 3 months, and the prevalence decreased with duration of diabetes. The antibodies were of the IgM class in 87% (13/15) of recent-onset Type 1 diabetes cases, but IgG-autoantibodies became more prevalent with increasing duration of diabetes. Three out of 347 (0.9%) Type 2 diabetic patients and 4 of 20 (20%) first-degree relatives of Type 1 diabetic patients had autoantibodies targeted against pancreatic exocrine cells. None of the patients with alcoholic pancreatitis or rheumatoid arthritis and none of the control subjects had these antibodies. Immunoblotting analysis and double-immunostaining demonstrated that the autoantibodies reacted with 40 kilodalton cytokeratin in pancreatic exocrine cell cytoplasm. The antibody was absorbed by the Triton X-100-insoluble fraction of pancreatic extract. These results indicate the presence of distinct autoantibodies to pancreatic exocrine cells in Type 1 diabetes. This suggests the provocative concept that the cytoskeletal system of pancreatic exocrine cells is involved in the pathogenetic process of Type 1 diabetes.
Article PDF
Similar content being viewed by others
References
Kobayashi T, Nakanishi K, Sugimoto T, Murase T, Kosaka K (1988) Histopathological changes of the pancreas in islet cell antibodies (ICA)-positive subjects before and after the clinical onset of insulin-dependent diabetes mellitus (IDDM). Diabetes 37: 24 (Abstract)
Kobayashi T, Nakanishi K (1987) Natural history of type I diabetes and slowly progressive IDDM: a histopathological study. J Jpn Diab Soc 30: 1152–1154
Nakanishi K, Kobayashi T, Sugimoto T, Murase T, Itoh T, Kosaka K (1988) Does pan-pancreatic involvement occur in IDDM? Diabetes Care 11: 100–101
Frier BM, Saunders JHB, Wormsley KG, Bouchier IAD (1976) Exocrine pancreatic function in juvenile-onset diabetes mellitus. Gut 17: 685–691
Lankisch PG, Manthey G, Otto J, Koop H, Talaulicar M, Willms B (1982) Exocrine pancreatic function in insulin-dependent diabetes mellitus. Digestion 25: 211–216
Thal AP, Murray MJ, Egner W (1959) Isoantibody formation in chronic pancreatic disease. Lancet I: 1128–1129
Lankisch PG, Koop H, Seelig R, Seelig HP (1981) Antinuclear and pancreatic acinar cell antibodies in pancreatic diseases. Digestion 21: 65–68
Lendrum R, Walker G (1975) Serum antibodies in human pancreatic disease. Gut 16: 365–371
Villavicencio E, Thurnau G, Goetz FC (1965) Isoantibodies to human pancreas: a preliminary report. Diabetes 14: 226–228
Ivarsson SA, Marner B, Lernmark Å, Nilsson KO (1988) Nonislet pancreatic autoantibodies in sibship with permanent neonatal insulin-dependent diabetes mellitus. Diabetes 37: 347–350
National Diabetes Data Group (1979) Classification and diagnosis of diabetes mellitus and other categories of glucose intolerance. Diabetes 28: 1039–1057
Steinert PM, Aynardi-Whitman M, Zackroff RV, Goldman RD (1982) Isolation and characterization of intermediate filaments. In: Wilson LA (ed) Methods and perspectives in cell biology, Part A. Academic Press, New York, pp 399–408
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Kobayashi T, Sugimoto S, Itoh T, Kosaka K, Tanaka T, Suwa S, Sato K, Tsuji K (1986) The prevalence of islet cell antibodies in Japanese insulin-dependent and non-insulin-dependent diabetic patients studied by indirect immunofluorescence and by a new method. Diabetes 35: 335–340
Kobayashi T, Itoh T, Kosaka K, Sato K, Tsuji K (1987) Time course of islet cell antibodies and B-cell function in non-insulindependent stage of type 1 diabetes. Diabetes 36: 510–517
Ohyama T, Kawamura K, Orimo H, Murakami M (1978) Radioimmunoassay method for measurement of quantity of pancreatic elastase-1 in human serum. J Clin Exp Med (Japan) 105: 91–97
Moll R, Franke WW, Schiller DL, Geiger B, Krepler R (1982) The catalog of human cytokeratins: patterns of expression in normal epithelia, tumors and cultured cells. Cell 31: 11–24
Tseng SCG, Jarvinen MJ, Nelson WG, Huang JW, Woodcock-Mitchell J, Sun TT (1982) Correlation of specific keratins with different types of epithelial differentiation: monoclonal antibody studies. Cell 30: 361–372
Lazarides E (1980) Intermediate filaments as mechanical integrators of cellular space. Nature 283: 249–256
Geiger B (1987) Intermediate filaments: looking for a function. Nature 329: 392–393
Kurki P, Virtanen I (1984) The detection of human antibodies against cytoskeletal components. J Immunol Methods 67: 209–223
Yoon JW, Austin M, Onodera T, Notkins AL (1979) Virus-induced diabetes mellitus: isolation of a virus from the pancreas of a child with diabetic ketoacidosis. N Engl J Med 300: 1173–1179
Nakanishi K, Kobayashi T, Sugimoto T, Murase T, Itoh T, Kosaka K, Hara M (1988) Inverse relationship between residual betacells and exocrine pancreas in insulin-dependent diabetics. Diab Res Clin Prac [Suppl.1]: S53 (Abstract)
Rayfield EJ, Seto Y (1978) Viruses and the pathogenesis of diabetes mellitus. Diabetes 27: 1126–1139
Lenk R, Penman S (1979) The cytoskeletal framework and poliovirus metabolism. Cell 16: 289–301
Sharpe AH, Chen LB, Fields BN (1982) The interaction of mammalian reoviruses with the cytoskeleton of monkey kidney CV-1 cells. Virology 120: 399–411
Murti KG, Goorha R (1983) Interaction of frog virus-3 with the cytoskeleton: altered organization of microtubules, intermediate filaments, and microfilaments. J Cell Biol 96: 1248–1257
Onodera T, Toniolo A, Ray UR, Jenson B, Knazek RA, Notkins AL (1981) Virus-induced diabetes mellitus: XX. Polyendocrinopathy and autoimmunity. J Exp Med 153: 1457–1473
Sotelo J, Gibbs CJ, Gajdusek DC (1980) Autoantibodies against axonal neurofilaments in patients with kuru and CreutzfeldtJakob disease. Science 210: 190–193
Rousset B, Vialettes B, Bernier-Valentin F, Vague P, Beylot M, Mornex R (1984) Anti-tubulin antibodies in recent onset Type 1 (insulin-dependent) diabetes mellitus: comparison with islet cell antibodies. Diabetologia 27: 427–432
Walker EJ, Jeffrey PD (1986) Polymyositis and molecular mimicry, a mechanism of autoimmunity. Lancet II: 605–607
Fujunami RS, Oldstone MBA, Wroblewska Z, Frankel ME, Koprowski H (1983) Molecular mimicry in virus infection: crossreaction of measles virus phosphoprotein or of herpes simplex virus protein with human intermediate filaments. Proc Natl Acad Sci USA 80: 2346–2350
Toh BH, Yildiz A, Sotelo J, Osung O, Holborow EJ, Kanakoudi F, Small JV (1979) Viral infections and IgM autoantibodies to cytoplasmic intermediate filaments. Clin Exp Immunol 37: 76–82
Bretherton L, Toh BH (1981) IgM autoantibody to intermediate filaments in infectious mononucleosis. J Clin Lab Immunol 5: 7–10
Pedersen JS, Toh BH, Locarnini SA, Gust ID, Shyamala GNS (1981) Autoantibody to intermediate filaments in viral hepatitis. Clin Immunol Immunopathol 21: 154–161
Vercammen M, Gorus F, Foriers A, Segers O, Somers G, Van de Winkel M, Pipeleers D (1989) Cell surface antibodies in Type 1 (insulin-dependent) diabetic patients. I. Presence of immunoglobulins M which bind to rat pituitary cells. II. Presence of immunoglobulins M which bind to lymphocytes. Diabetologia 32: 611–617
Segers O, Gorus F, Somers G, Van de Winkel M, Vercammen M, Pipeleers D (1989) Cell surface antibodies in Type 1 (insulin-dependent) diabetic patients. II. Presence of immunoglobulins M which bind to lymphocytes. Diabetologia 32: 618–623
Nakamura M, Burastero SE, Notkins AL, Casali P (1988) Human monoclonal rheumatoid factor-like antibodies from CDS (Leu-1)+ B cells are polyreactive. J Immunol 140: 4180–4186
Casali P, Notkins AL (1989) CD5+ B lymphocytes, polyreactive antibodies and the human B-cell repertoire. Immunolo Today 10: 364–368
Young BJJ, Mallya RK, Leslie RDG, Clark CJM, Hamblin TJ (1979) Anti-keratin antibodies in rheumatoid arthritis. Br Med J 2: 97–99
Kataaha PK, Mortazavi-Milani SM, Russell G, Holborow EJ (1985) Anti-intermediate filament antibodies, antikeratin antibody, and antiperinuclear factor in rheumatoid arthritis and infectious mononucleosis. Ann Rheum Dis 44: 446–449
Ulrich K, Schubart K, Fields KL (1984) Identification of a calcium-regulated insulinoma cell phosphoprotein as an islet cell keratin. J Cell Biol 98: 1001–1009
Dulbecco R, Unger M, Bologna M, Battifora H, Syka P, Okada S (1981) Cross-reactivity between Thy-1 and a component of intermediate filaments demonstrated using a monoclonal antibody. Nature 292: 772–774
Dales S, Fujinami RS, Oldstone MBA (1983) Serologic relatedness between Thy-1.2 and actin revealed by monoclonal antibody. J Immunol 131: 1332–1338
Diaz LA, Sampaio SAP, Martins CR, Rivitti EA, Macca ML, Roscoe JT, Takahashi Y, Labib RS, Patel HP, Mutasim DF, Dugan EM, Anhalt GJ (1987) An autoantibody in pemphigus serum, specific for the 59 kD keratin, selectively binds the surface of keratinocytes: evidence for an extracellular keratin domain. J Invest Dermatol 89: 287–295
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kobayashi, T., Nakanishi, K., Kajio, H. et al. Pancreatic cytokeratin: an antigen of pancreatic exocrine cell autoantibodies in Type 1 (insulin-dependent) diabetes mellitus. Diabetologia 33, 363–370 (1990). https://doi.org/10.1007/BF00404641
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00404641