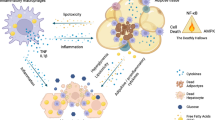
Abstract
Obesity is an established risk factor in the pathogenesis of insulin resistance, type 2 diabetes mellitus and cardiovascular disease; all components that are part of the metabolic syndrome. Traditionally, insulin resistance has been defined in a glucocentric perspective. However, elevated systemic levels of fatty acids are now considered significant contributors towards the pathophysiological aspects associated with the syndrome. An overaccumulation of unoxidized long-chain fatty acids can saturate the storage capacity of adipose tissue, resulting in a lipid ‘spill over’ to non-adipose tissues, such as the liver, muscle, heart, and pancreatic-islets. Under these circumstances, such ectopic lipid deposition can have deleterious effects. The excess lipids are driven into alternative non-oxidative pathways, which result in the formation of reactive lipid moieties that promote metabolically relevant cellular dysfunction (lipotoxicity) and programmed cell-death (lipoapoptosis). Here, we focus on how both of these processes affect metabolically significant cell-types and highlight how lipotoxicity and sequential lipoapoptosis are as major mediators of insulin resistance, diabetes and cardiovascular disease.



Similar content being viewed by others
Abbreviations
- T2DM:
-
Type 2 diabetes mellitus
- CVD:
-
Cardiovascular disease
- TG:
-
Triglyceride/triacylglycerol
- FFA:
-
Free fatty acid
- GLUT4:
-
Glucose transporter-4
- NO:
-
Nitric oxide
- AMPK:
-
Adenosine monophosphate-activated protein kinase
- CoA:
-
Coenzyme-A
- SREBP-1c:
-
Sterol regulatory element binding protein-1c
- PPAR-γ2:
-
Peroxisome proliferator-activated receptor
- ACC:
-
Acetyl coenzyme A carboxylase
- FAS:
-
Fatty acid synthetase
- GPAT:
-
Glycerol-3-phosphate acyl transferase
- CPT-1:
-
Carnitine palmityl transferase-1
- ACO:
-
Fatty acyl-CoA oxidase
- PGC-1α:
-
Peroxisome proliferator-activated receptor-γ coactivator-1α
- UCP-2:
-
Uncoupling protein-2
- VLDLs:
-
Very low-density lipoproteins
- ROS:
-
Reactive oxygen species
- ER:
-
Endoplasmic reticulum
- SPT-1:
-
Serine palmitoyl transferase
- Bcl2 :
-
B-cell lymphoma 2
- Bad:
-
Bcl2-antagonist of cell death
- Bax:
-
Bcl2-associated X protein
- Bid:
-
BH3 interacting domain death agonist
- Bim:
-
Bcl2-like 11
- NF-κB:
-
Nuclear factor-κB
- iNOS:
-
Inducible nitric oxide synthase
- ACS:
-
Acyl CoA synthase
- ECM:
-
Extracellular matrix
- FAT-ATTAC:
-
Fat apoptosis through targeted activation of caspase-8
- FKBP:
-
Peptidyl-prolyl cis–trans isomerase
- AICAR:
-
5-Amino 4-imidazolecarboxamide riboside
- MCD:
-
Malonyl CoA decarboxylase
References
Reaven GM (1988) Banting lecture 1988. Role of insulin resistance in human disease. Diabetes 37:1595–1607. doi:10.2337/diabetes.37.12.1595
Randle PJ, Garland PB, Hales CN et al (1963) The glucose fatty-acid cycle. Its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus. Lancet 1:785–789. doi:10.1016/S0140-6736(63)91500-9
McGarry JD (1992) What if Minkowski had been ageusic? An alternative angle on diabetes. Science 258:766–770. doi:10.1126/science.1439783
Shulman GI (2000) Cellular mechanisms of insulin resistance. J Clin Invest 106:171–176. doi:10.1172/JCI10583
Boden G, Cheung P, Stein TP et al (2002) FFA cause hepatic insulin resistance by inhibiting insulin suppression of glycogenolysis. Am J Physiol Endocrinol Metab 283:E12–E19
Charles MA, Eschwege E, Thibult N et al (1997) The role of non-esterified fatty acids in the deterioration of glucose tolerance in caucasian subjects: results of the Paris prospective study. Diabetologia 40:1101–1106. doi:10.1007/s001250050793
Schulz LO, Bennett PH, Ravussin E et al (2006) Effects of traditional and western environments on prevalence of type 2 diabetes in Pima Indians in Mexico and the US. Diabetes Care 29:1866–1871. doi:10.2337/dc06-0138
Lee Y, Hirose H, Ohneda M et al (1994) Beta-cell lipotoxicity in the pathogenesis of non-insulin-dependent diabetes mellitus of obese rats: impairment in adipocyte-beta-cell relationships. Proc Natl Acad Sci USA 91:10878–10882. doi:10.1073/pnas.91.23.10878
Unger RH (2003) Lipid overload and overflow: metabolic trauma and the metabolic syndrome. Trends Endocrinol Metab 14:398–403. doi:10.1016/j.tem.2003.09.008
Unger RH, Orci L (2001) Diseases of liporegulation: new perspective on obesity and related disorders. FASEB J 15:312–321. doi:10.1096/fj.00-0590
Neel JV (1962) Diabetes mellitus: a “thrifty” genotype rendered detrimental by “progress”? Am J Hum Genet 14:353–362
Simha V, Garg A (2006) Lipodystrophy: lessons in lipid and energy metabolism. Curr Opin Lipidol 17:162–169
Zhang Y, Proenca R, Maffei M et al (1994) Positional cloning of the mouse obese gene and its human homologue. Nature 372:425–432. doi:10.1038/372425a0
Halaas JL, Gajiwala KS, Maffei M et al (1995) Weight-reducing effects of the plasma protein encoded by the obese gene. Science 269:543–546. doi:10.1126/science.7624777
Campfield LA, Smith FJ, Guisez Y et al (1995) Recombinant mouse OB protein: evidence for a peripheral signal linking adiposity and central neural networks. Science 269:546–549. doi:10.1126/science.7624778
Shimabukuro M, Koyama K, Chen G et al (1997) Direct antidiabetic effect of leptin through triglyceride depletion of tissues. Proc Natl Acad Sci USA 94:4637–4641. doi:10.1073/pnas.94.9.4637
Wang MY, Koyama K, Shimabukuro M et al (1998) OB-Rb gene transfer to leptin-resistant islets reverses diabetogenic phenotype. Proc Natl Acad Sci USA 95:714–718. doi:10.1073/pnas.95.2.714
Lee Y, Wang MY, Kakuma T et al (2001) Liporegulation in diet-induced obesity. The antisteatotic role of hyperleptinemia. J Biol Chem 276:5629–5635. doi:10.1074/jbc.M008553200
Zhou YT, Grayburn P, Karim A et al (2000) Lipotoxic heart disease in obese rats: implications for human obesity. Proc Natl Acad Sci USA 97:1784–1789. doi:10.1073/pnas.97.4.1784
Muller-Wieland D, Kotzka J (2005) Correction of insulin resistance and the metabolic syndrome. Handb Exp Pharmacol 170:591–617
Scherer PE, Williams S, Fogliano M et al (1995) A novel serum protein similar to C1q, produced exclusively in adipocytes. J Biol Chem 270:26746–26749. doi:10.1074/jbc.270.45.26746
Kim JY, van de Wall E, Laplante M et al (2007) Obesity-associated improvements in metabolic profile through expansion of adipose tissue. J Clin Invest 117:2621–2637. doi:10.1172/JCI31021
Combs TP, Wagner JA, Berger J et al (2002) Induction of adipocyte complement-related protein of 30 kilodaltons by PPARgamma agonists: a potential mechanism of insulin sensitization. Endocrinology 143:998–1007. doi:10.1210/en.143.3.998
Wang ZV, Scherer PE (2008) Adiponectin, cardiovascular function, and hypertension. Hypertension 51:8–14. doi:10.1161/HYPERTENSIONAHA.107.099424
Tomas E, Tsao TS, Saha AK et al (2002) Enhanced muscle fat oxidation and glucose transport by ACRP30 globular domain: acetyl-CoA carboxylase inhibition and AMP-activated protein kinase activation. Proc Natl Acad Sci USA 99:16309–16313. doi:10.1073/pnas.222657499
Yamauchi T, Kamon J, Waki H et al (2001) The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat Med 7:941–946. doi:10.1038/90984
Yamauchi T, Kamon J, Minokoshi Y et al (2002) Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat Med 8:1288–1295. doi:10.1038/nm788
Brown MS, Goldstein JL (1998) Sterol regulatory element binding proteins (SREBPs): controllers of lipid synthesis and cellular uptake. Nutr Rev 56:S1–S3 discussion S54–75
Wang X, Sato R, Brown MS et al (1994) SREBP-1, a membrane-bound transcription factor released by sterol-regulated proteolysis. Cell 77:53–62. doi:10.1016/0092-8674(94)90234-8
Kakuma T, Wang ZW, Pan W et al (2000) Role of leptin in peroxisome proliferator-activated receptor gamma coactivator-1 expression. Endocrinology 141:4576–4582. doi:10.1210/en.141.12.4576
Zhou YT, Shimabukuro M, Koyama K et al (1997) Induction by leptin of uncoupling protein-2 and enzymes of fatty acid oxidation. Proc Natl Acad Sci USA 94:6386–6390. doi:10.1073/pnas.94.12.6386
Hardie DG, Pan DA (2002) Regulation of fatty acid synthesis and oxidation by the AMP-activated protein kinase. Biochem Soc Trans 30:1064–1070. doi:10.1042/BST0301064
McGarry JD, Mannaerts GP, Foster DW (1977) A possible role for malonyl-CoA in the regulation of hepatic fatty acid oxidation and ketogenesis. J Clin Invest 60:265–270. doi:10.1172/JCI108764
McGarry JD (2002) Banting lecture 2001: dysregulation of fatty acid metabolism in the etiology of type 2 diabetes. Diabetes 51:7–18. doi:10.2337/diabetes.51.1.7
Lee Y, Yu X, Gonzales F et al (2002) PPAR alpha is necessary for the lipopenic action of hyperleptinemia on white adipose and liver tissue. Proc Natl Acad Sci USA 99:11848–11853. doi:10.1073/pnas.182420899
Spiegelman BM (2007) Transcriptional control of mitochondrial energy metabolism through the PGC1 coactivators. Novartis Found Symp 287:60–63. doi:10.1002/9780470725207.ch5 discussion 63–69
Moitra J, Mason MM, Olive M et al (1998) Life without white fat: a transgenic mouse. Genes Dev 12:3168–3181. doi:10.1101/gad.12.20.3168
Florant GL, Porst H, Peiffer A et al (2004) Fat-cell mass, serum leptin and adiponectin changes during weight gain and loss in yellow-bellied marmots (Marmota flaviventris). J Comp Physiol [B] 174:633–639. doi:10.1007/s00360-004-0454-0
Mason TM (1998) The role of factors that regulate the synthesis and secretion of very-low-density lipoprotein by hepatocytes. Crit Rev Clin Lab Sci 35:461–487. doi:10.1080/10408369891234246
Shimabukuro M, Zhou YT, Levi M et al (1998) Fatty acid-induced beta cell apoptosis: a link between obesity and diabetes. Proc Natl Acad Sci USA 95:2498–2502. doi:10.1073/pnas.95.5.2498
Szczepaniak LS, Victor RG, Orci L et al (2007) Forgotten but not gone: the rediscovery of fatty heart, the most common unrecognized disease in America. Circ Res 101:759–767. doi:10.1161/CIRCRESAHA.107.160457
Unger RH, Zhou YT, Orci L (1999) Regulation of fatty acid homeostasis in cells: novel role of leptin. Proc Natl Acad Sci USA 96:2327–2332. doi:10.1073/pnas.96.5.2327
Unger RH (2005) Longevity, lipotoxicity and leptin: the adipocyte defense against feasting and famine. Biochimie 87:57–64. doi:10.1016/j.biochi.2004.11.014
Zhou YT, Shimabukuro M, Lee Y et al (1998) Enhanced de novo lipogenesis in the leptin-unresponsive pancreatic islets of prediabetic Zucker diabetic fatty rats: role in the pathogenesis of lipotoxic diabetes. Diabetes 47:1904–1908. doi:10.2337/diabetes.47.12.1904
Quon MJ, Butte AJ, Zarnowski MJ et al (1994) Insulin receptor substrate 1 mediates the stimulatory effect of insulin on GLUT4 translocation in transfected rat adipose cells. J Biol Chem 269:27920–27924
McGarry JD, Brown NF (1997) The mitochondrial carnitine palmitoyltransferase system. From concept to molecular analysis. Eur J Biochem 244:1–14. doi:10.1111/j.1432-1033.1997.00001.x
Summers SA, Garza LA, Zhou H et al (1998) Regulation of insulin-stimulated glucose transporter GLUT4 translocation and Akt kinase activity by ceramide. Mol Cell Biol 18:5457–5464
Robertson RP, Harmon JS (2006) Diabetes, glucose toxicity, and oxidative stress: a case of double jeopardy for the pancreatic islet beta cell. Free Radic Biol Med 41:177–184. doi:10.1016/j.freeradbiomed.2005.04.030
Maechler P, Jornot L, Wollheim CB (1999) Hydrogen peroxide alters mitochondrial activation and insulin secretion in pancreatic beta cells. J Biol Chem 274:27905–27913. doi:10.1074/jbc.274.39.27905
Tiedge M, Lortz S, Drinkgern J et al (1997) Relation between antioxidant enzyme gene expression and antioxidative defense status of insulin-producing cells. Diabetes 46:1733–1742. doi:10.2337/diabetes.46.11.1733
Unger RH, Zhou YT (2001) Lipotoxicity of beta-cells in obesity and in other causes of fatty acid spillover. Diabetes 50(Suppl 1):S118–S121. doi:10.2337/diabetes.50.2007.S118
Ohneda M, Inman LR, Unger RH (1995) Caloric restriction in obese pre-diabetic rats prevents beta-cell depletion, loss of beta-cell GLUT 2 and glucose incompetence. Diabetologia 38:173–179. doi:10.1007/BF00400091
Ribaux PG, Iynedjian PB (2003) Analysis of the role of protein kinase B (cAKT) in insulin-dependent induction of glucokinase and sterol regulatory element-binding protein 1 (SREBP1) mRNAs in hepatocytes. Biochem J 376:697–705. doi:10.1042/BJ20031287
Koshkin V, Wang X, Scherer PE et al (2003) Mitochondrial functional state in clonal pancreatic beta-cells exposed to free fatty acids. J Biol Chem 278:19709–19715. doi:10.1074/jbc.M209709200
Carlsson C, Borg LA, Welsh N (1999) Sodium palmitate induces partial mitochondrial uncoupling and reactive oxygen species in rat pancreatic islets in vitro. Endocrinology 140:3422–3428. doi:10.1210/en.140.8.3422
Rakatzi I, Mueller H, Ritzeler O et al (2004) Adiponectin counteracts cytokine- and fatty acid-induced apoptosis in the pancreatic beta-cell line INS-1. Diabetologia 47:249–258. doi:10.1007/s00125-003-1293-3
Gu W, Li X, Liu C et al (2006) Globular adiponectin augments insulin secretion from pancreatic islet beta cells at high glucose concentrations. Endocrine 30:217–221. doi:10.1385/ENDO:30:2:217
Huypens P, Moens K, Heimberg H et al (2005) Adiponectin-mediated stimulation of AMP-activated protein kinase (AMPK) in pancreatic beta cells. Life Sci 77:1273–1282. doi:10.1016/j.lfs.2005.03.008
Staiger K, Stefan N, Staiger H et al (2005) Adiponectin is functionally active in human islets but does not affect insulin secretory function or beta-cell lipoapoptosis. J Clin Endocrinol Metab 90:6707–6713. doi:10.1210/jc.2005-0467
Huypens PR (2007) Leptin and adiponectin regulate compensatory beta cell growth in accordance to overweight. Med Hypotheses 68:1134–1137. doi:10.1016/j.mehy.2006.09.046
Shimomura I, Bashmakov Y, Ikemoto S et al (1999) Insulin selectively increases SREBP-1c mRNA in the livers of rats with streptozotocin-induced diabetes. Proc Natl Acad Sci USA 96:13656–13661. doi:10.1073/pnas.96.24.13656
Shimomura I, Hammer RE, Ikemoto S et al (1999) Leptin reverses insulin resistance and diabetes mellitus in mice with congenital lipodystrophy. Nature 401:73–76. doi:10.1038/43448
Ma K, Cabrero A, Saha PK et al (2002) Increased beta-oxidation but no insulin resistance or glucose intolerance in mice lacking adiponectin. J Biol Chem 277:34658–34661. doi:10.1074/jbc.C200362200
Wang MY, Orci L, Ravazzola M et al (2005) Fat storage in adipocytes requires inactivation of leptin’s paracrine activity: implications for treatment of human obesity. Proc Natl Acad Sci USA 102:18011–18016. doi:10.1073/pnas.0509001102
Chiu HC, Kovacs A, Ford DA et al (2001) A novel mouse model of lipotoxic cardiomyopathy. J Clin Invest 107:813–822. doi:10.1172/JCI10947
Obeid LM, Linardic CM, Karolak LA et al (1993) Programmed cell death induced by ceramide. Science 259:1769–1771. doi:10.1126/science.8456305
Karaskov E, Scott C, Zhang L et al (2006) Chronic palmitate but not oleate exposure induces endoplasmic reticulum stress, which may contribute to INS-1 pancreatic beta-cell apoptosis. Endocrinology 147:3398–3407. doi:10.1210/en.2005-1494
Shimabukuro M, Higa M, Zhou YT et al (1998) Lipoapoptosis in beta-cells of obese prediabetic fa/fa rats. Role of serine palmitoyltransferase overexpression. J Biol Chem 273:32487–32490. doi:10.1074/jbc.273.49.32487
Weiss B, Stoffel W (1997) Human and murine serine-palmitoyl-CoA transferase—cloning, expression and characterization of the key enzyme in sphingolipid synthesis. Eur J Biochem 249:239–247. doi:10.1111/j.1432-1033.1997.00239.x
Maedler K, Oberholzer J, Bucher P et al (2003) Monounsaturated fatty acids prevent the deleterious effects of palmitate and high glucose on human pancreatic beta-cell turnover and function. Diabetes 52:726–733. doi:10.2337/diabetes.52.3.726
Kulik G, Klippel A, Weber MJ (1997) Antiapoptotic signalling by the insulin-like growth factor I receptor, phosphatidylinositol 3-kinase, and Akt. Mol Cell Biol 17:1595–1606
Holland WL, Summers SA (2008) Sphingolipids, insulin resistance, and metabolic disease: new insights from in vivo manipulation of sphingolipid metabolism. Endocr Rev 29:381–402. doi:10.1210/er.2007-0025
Datta SR, Dudek H, Tao X et al (1997) Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 91:231–241. doi:10.1016/S0092-8674(00)80405-5
El-Assaad W, Buteau J, Peyot ML et al (2003) Saturated fatty acids synergize with elevated glucose to cause pancreatic beta-cell death. Endocrinology 144:4154–4163. doi:10.1210/en.2003-0410
Briaud I, Harmon JS, Kelpe CL et al (2001) Lipotoxicity of the pancreatic beta-cell is associated with glucose-dependent esterification of fatty acids into neutral lipids. Diabetes 50:315–321. doi:10.2337/diabetes.50.2.315
Shimabukuro M, Ohneda M, Lee Y et al (1997) Role of nitric oxide in obesity-induced beta cell disease. J Clin Invest 100:290–295. doi:10.1172/JCI119534
Schonfeld P, Wojtczak L (2007) Fatty acids decrease mitochondrial generation of reactive oxygen species at the reverse electron transport but increase it at the forward transport. Biochim Biophys Acta 1767:1032–1040. doi:10.1016/j.bbabio.2007.04.005
Gudz TI, Tserng KY, Hoppel CL (1997) Direct inhibition of mitochondrial respiratory chain complex III by cell-permeable ceramide. J Biol Chem 272:24154–24158. doi:10.1074/jbc.272.39.24154
Pi J, Bai Y, Zhang Q et al (2007) Reactive oxygen species as a signal in glucose-stimulated insulin secretion. Diabetes 56:1783–1791. doi:10.2337/db06-1601
Cnop M, Welsh N, Jonas JC et al (2005) Mechanisms of pancreatic beta-cell death in type 1 and type 2 diabetes: many differences, few similarities. Diabetes 54(Suppl 2):S97–S107. doi:10.2337/diabetes.54.suppl_2.S97
Lameloise N, Muzzin P, Prentki M et al (2001) Uncoupling protein 2: a possible link between fatty acid excess and impaired glucose-induced insulin secretion? Diabetes 50:803–809. doi:10.2337/diabetes.50.4.803
Joseph JW, Koshkin V, Saleh MC et al (2004) Free fatty acid-induced beta-cell defects are dependent on uncoupling protein 2 expression. J Biol Chem 279:51049–51056. doi:10.1074/jbc.M409189200
Shimabukuro M, Wang MY, Zhou YT et al (1998) Protection against lipoapoptosis of beta cells through leptin-dependent maintenance of Bcl-2 expression. Proc Natl Acad Sci USA 95:9558–9561. doi:10.1073/pnas.95.16.9558
Cheng L, Ding G, Qin Q et al (2004) Cardiomyocyte-restricted peroxisome proliferator-activated receptor-delta deletion perturbs myocardial fatty acid oxidation and leads to cardiomyopathy. Nat Med 10:1245–1250. doi:10.1038/nm1116
Park TS, Hu Y, Noh HL et al (2008) Ceramide is a cardiotoxin in lipotoxic cardiomyopathy. J Lipid Res 49:2101–2112. doi:10.1194/jlr.M800147-JLR200
Hickson-Bick DL, Buja LM, McMillin JB (2000) Palmitate-mediated alterations in the fatty acid metabolism of rat neonatal cardiac myocytes. J Mol Cell Cardiol 32:511–519. doi:10.1006/jmcc.1999.1098
Sharma S, Adrogue JV, Golfman L et al (2004) Intramyocardial lipid accumulation in the failing human heart resembles the lipotoxic rat heart. FASEB J 18:1692–1700. doi:10.1096/fj.04-2263com
Kankaanpaa M, Lehto HR, Parkka JP et al (2006) Myocardial triglyceride content and epicardial fat mass in human obesity: relationship to left ventricular function and serum free fatty acid levels. J Clin Endocrinol Metab 91:4689–4695. doi:10.1210/jc.2006-0584
Spalding KL, Arner E, Westermark PO et al (2008) Dynamics of fat cell turnover in humans. Nature 453:783–787. doi:10.1038/nature06902
Khan T, Muise ES, Iyengar P et al. (2009) Metabolic dysregulation and adipose tissue fibrosis: the role of collagen VI. Mol Cell Biol 29:1575–1591
Demeulemeester D, Collen D, Lijnen HR (2005) Effect of matrix metalloproteinase inhibition on adipose tissue development. Biochem Biophys Res Commun 329:105–110. doi:10.1016/j.bbrc.2005.01.103
Strissel KJ, Stancheva Z, Miyoshi H et al (2007) Adipocyte death, adipose tissue remodeling, and obesity complications. Diabetes 56:2910–2918. doi:10.2337/db07-0767
Duffield JS (2003) The inflammatory macrophage: a story of Jekyll and Hyde. Clin Sci (Lond) 104:27–38. doi:10.1042/CS20020240
Pajvani UB, Trujillo ME, Combs TP et al (2005) Fat apoptosis through targeted activation of caspase 8: a new mouse model of inducible and reversible lipoatrophy. Nat Med 11:797–803. doi:10.1038/nm1262
Saha AK, Ruderman NB (2003) Malonyl-CoA and AMP-activated protein kinase: an expanding partnership. Mol Cell Biochem 253:65–70. doi:10.1023/A:1026053302036
Higa M, Zhou YT, Ravazzola M et al (1999) Troglitazone prevents mitochondrial alterations, beta cell destruction, and diabetes in obese prediabetic rats. Proc Natl Acad Sci USA 96:11513–11518. doi:10.1073/pnas.96.20.11513
Acknowledgments
We kindly thank Kate McCorkle for assistance with the figures. Our research is supported by NIH grants R01-DK55758 and R01-CA112023 (PES) and a JDRF post-doctoral fellowship 3-2008-130 (CMK).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kusminski, C.M., Shetty, S., Orci, L. et al. Diabetes and apoptosis: lipotoxicity. Apoptosis 14, 1484–1495 (2009). https://doi.org/10.1007/s10495-009-0352-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-009-0352-8