Abstract
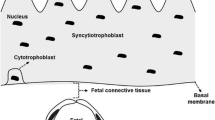
By its location between maternal and fetal bloodstreams the human placenta not only handles the materno-fetal transport of nutrients and gases, but may also be exposed to intrauterine conditions adversely affecting placental and fetal development. Such adverse conditions exist in pregnancies complicated by gestational diabetes mellitus (GDM), and have been associated with alterations in placental anatomy and physiology. These alterations are mainly based on changes on the micro-anatomical and/or even molecular level including aberrant villous vascularization, a disbalance of vasoactive molecules, and enhanced oxidative stress. The consequence thereof may be impaired fetal oxygenation and changes in transplacental nutrient supply. Although transplacental glucose flux is flow limited and independent of glucose transporter availability, transport of essential and nonessential amino acids and expression of genes involved in lipid transport and metabolism are significantly affected by GDM.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Ben-Haroush A, Yogev Y, Hod M. Epidemiology of gestational diabetes mellitus and its association with Type 2 diabetes. Diabet Med. 2004;21:103–13.
Radaelli T, Varastehpour A, Catalano P, et al. Gestational diabetes induces placental genes for chronic stress and inflammatory pathways. Diabetes. 2003;52:2951–8.
Segregur J, Bukovic D, Milinovic D, et al. Fetal macrosomia in pregnant women with gestational diabetes. Coll Antropol. 2009;33:1121–7.
Kuhl C, Hornnes PJ, Andersen O. Etiology and pathophysiology of gestational diabetes mellitus. Diabetes. 1985;34 Suppl 2:66–70.
Kautzky-Willer A, Prager R, Waldhausl W, et al. Pronounced insulin resistance and inadequate beta-cell secretion characterize lean gestational diabetes during and after pregnancy. Diabetes Care. 1997;20:1717–23.
Xiang AH, Peters RK, Trigo E, et al. Multiple metabolic defects during late pregnancy in women at high risk for type 2 diabetes. Diabetes. 1999;48:848–54.
Taricco E, Radaelli T, Rossi G, et al. Effects of gestational diabetes on fetal oxygen and glucose levels in vivo. Bjog. 2009;116:1729–35.
Daskalakis G, Marinopoulos S, Krielesi V, et al. Placental pathology in women with gestational diabetes. Acta Obstet Gynecol Scand. 2008;87:403–7.
Madazli R, Tuten A, Calay Z, et al. The incidence of placental abnormalities, maternal and cord plasma malondialdehyde and vascular endothelial growth factor levels in women with gestational diabetes mellitus and nondiabetic controls. Gynecol Obstet Invest. 2008;65:227–32.
Schafer-Graf UM, Dupak J, Vogel M, et al. Hyperinsulinism, neonatal obesity and placental immaturity in infants born to women with one abnormal glucose tolerance test value. J Perinat Med. 1998;26:27–36.
Bartha JL, Martinez-Del-Fresno P, Comino-Delgado R. Gestational diabetes mellitus diagnosed during early pregnancy. Am J Obstet Gynecol. 2000;182:346–50.
Meyer WJ, Carbone J, Gauthier DW, et al. Early gestational glucose screening and gestational diabetes. J Reprod Med. 1996;41:675–9.
Cetin I, de Santis MS, Taricco E, et al. Maternal and fetal amino acid concentrations in normal pregnancies and in pregnancies with gestational diabetes mellitus. Am J Obstet Gynecol. 2005;192:610–7.
Taricco E, Radaelli T, Nobile de Santis MS, et al. Foetal and placental weights in relation to maternal characteristics in gestational diabetes. Placenta. 2003;24:343–7.
Kucuk M, Doymaz F. Placental weight and placental weight-to-birth weight ratio are increased in diet- and exercise-treated gestational diabetes mellitus subjects but not in subjects with one abnormal value on 100-g oral glucose tolerance test. J Diabetes Complications. 2009;23:25–31.
Lao TT, Lee CP, Wong WM. Placental weight to birthweight ratio is increased in mild gestational glucose intolerance. Placenta. 1997;18:227–30.
Chan KK, Ho LF, Lao TT. Nutritional intake and placental size in gestational diabetic pregnancies–a preliminary observation. Placenta. 2003;24:985–8.
Pathak S, Hook E, Hackett G, et al. Cord coiling, umbilical cord insertion and placental shape in an unselected cohort delivering at term: relationship with common obstetric outcomes. Placenta. 2010;31:963–8.
Calderon IM, Damasceno DC, Amorin RL, et al. Morphometric study of placental villi and vessels in women with mild hyperglycemia or gestational or overt diabetes. Diabetes Res Clin Pract. 2007;78:65–71.
al-Okail MS, al-Attas OS. Histological changes in placental syncytiotrophoblasts of poorly controlled gestational diabetic patients. Endocr J. 1994;41:355–60.
Nadra K, Quignodon L, Sardella C, et al. PPARgamma in placental angiogenesis. Endocrinology. 2010;151:4969–81.
Reynolds LP, Redmer DA. Angiogenesis in the placenta. Biol Reprod. 2001;64:1033–40.
Khaliq A, Li XF, Shams M, et al. Localisation of placenta growth factor (PIGF) in human term placenta. Growth Factors. 1996;13:243–50. color plates I-II,pre bk cov.
Lang I, Pabst MA, Hiden U, et al. Heterogeneity of microvascular endothelial cells isolated from human term placenta and macrovascular umbilical vein endothelial cells. Eur J Cell Biol. 2003;82:163–73.
Murthi P, Hiden U, Rajaraman G, et al. Novel homeobox genes are differentially expressed in placental microvascular endothelial cells compared with macrovascular cells. Placenta. 2008;29:624–30.
Murthi P, So M, Gude NM, et al. Homeobox genes are differentially expressed in macrovascular human umbilical vein endothelial cells and microvascular placental endothelial cells. Placenta. 2007;28:219–23.
Grissa O, Yessoufou A, Mrisak I, et al. Growth factor concentrations and their placental mRNA expression are modulated in gestational diabetes mellitus: possible interactions with macrosomia. BMC Pregnancy Childbirth. 2010;10:7.
Hill DJ, Tevaarwerk GJ, Caddell C, et al. Fibroblast growth factor 2 is elevated in term maternal and cord serum and amniotic fluid in pregnancies complicated by diabetes: relationship to fetal and placental size. J Clin Endocrinol Metab. 1995;80:2626–32.
Holdsworth-Carson SJ, Lim R, Mitton A, et al. Peroxisome proliferator-activated receptors are altered in pathologies of the human placenta: gestational diabetes mellitus, intrauterine growth restriction and preeclampsia. Placenta. 2010;31:222–9.
• Acosta JC, Haas DM, Saha CK et al (2011) Gestational diabetes mellitus alters maternal and neonatal circulating endothelial progenitor cell subsets. Am J Obstet Gynecol 204: 254 e258-254 e215. Circulating endothelial progenitor cells are reduced in an intrauterine GDM environment. This may contribute to endothelial dysfunction in placenta and program the offspring for later disease associated with impaired endothelial function.
Estes ML, Mund JA, Mead LE, et al. Application of polychromatic flow cytometry to identify novel subsets of circulating cells with angiogenic potential. Cytometry A. 2010;77:831–9.
Jirkovska M, Kubinova L, Janacek J, et al. Topological properties and spatial organization of villous capillaries in normal and diabetic placentas. J Vasc Res. 2002;39:268–78.
Babawale MO, Lovat S, Mayhew TM, et al. Effects of gestational diabetes on junctional adhesion molecules in human term placental vasculature. Diabetologia. 2000;43:1185–96.
Fadda GM, D'Antona D, Ambrosini G, et al. Placental and fetal pulsatility indices in gestational diabetes mellitus. J Reprod Med. 2001;46:365–70.
Brown MA, North L, Hargood J. Uteroplacental Doppler ultrasound in routine antenatal care. Aust N Z J Obstet Gynaecol. 1990;30:303–7.
Pietryga M, Brazert J, Wender-Ozegowska E, et al. Placental Doppler velocimetry in gestational diabetes mellitus. J Perinat Med. 2006;34:108–10.
Reisenberger K, Egarter C, Kapiotis S, et al. Transfer of erythropoietin across the placenta perfused in vitro. Obstet Gynecol. 1997;89:738–42.
Leushner JR, Tevaarwerk GJ, Clarson CL, et al. Analysis of the collagens of diabetic placental villi. Cell Mol Biol. 1986;32:27–35.
Stanley JL, Cheung CC, Rueda-Clausen CF, et al. Effect of gestational diabetes on maternal artery function. Reprod Sci. 2011;18:342–52.
Bobadilla RA, van Bree R, Vercruysse L, et al. Placental effects of systemic tumour necrosis factor-alpha in an animal model of gestational diabetes mellitus. Placenta. 2010;31:1057–63.
Mildenberger E, Biesel B, Siegel G, et al. Nitric oxide and endothelin in oxygen-dependent regulation of vascular tone of human umbilical vein. Am J Physiol Heart Circ Physiol. 2003;285:H1730–7.
Boura AL, Walters WA, Read MA, et al. Autacoids and control of human placental blood flow. Clin Exp Pharmacol Physiol. 1994;21:737–48.
San Martin R, Sobrevia L. Gestational diabetes and the adenosine/L-arginine/nitric oxide (ALANO) pathway in human umbilical vein endothelium. Placenta. 2006;27:1–10.
Schonfelder G, John M, Hopp H, et al. Expression of inducible nitric oxide synthase in placenta of women with gestational diabetes. Faseb J. 1996;10:777–84.
Figueroa R, Martinez E, Fayngersh RP, et al. Alterations in relaxation to lactate and H(2)O(2) in human placental vessels from gestational diabetic pregnancies. Am J Physiol Heart Circ Physiol. 2000;278:H706–13.
Sobrevia L, Cesare P, Yudilevich DL, et al. Diabetes-induced activation of system y + and nitric oxide synthase in human endothelial cells: association with membrane hyperpolarization. J Physiol. 1995;489(Pt 1):183–92.
Sobrevia L, Yudilevich DL, Mann GE. Elevated D-glucose induces insulin insensitivity in human umbilical endothelial cells isolated from gestational diabetic pregnancies. J Physiol. 1998;506(Pt 1):219–30.
Vasquez G, Sanhueza F, Vasquez R, et al. Role of adenosine transport in gestational diabetes-induced L-arginine transport and nitric oxide synthesis in human umbilical vein endothelium. J Physiol. 2004;560:111–22.
De Vriese AS, Verbeuren TJ, Van de Voorde J, et al. Endothelial dysfunction in diabetes. Br J Pharmacol. 2000;130:963–74.
Tooke JE, Goh KL. Endotheliopathy precedes type 2 diabetes. Diabetes Care. 1998;21:2047–9.
Sobrevia L, Mann GE. Dysfunction of the endothelial nitric oxide signalling pathway in diabetes and hyperglycaemia. Exp Physiol. 1997;82:423–52.
• Sobrevia L, Gonzalez M (2009) A role for insulin on L-arginine transport in fetal endothelial dysfunction in hyperglycaemia. Curr Vasc Pharmacol 7: 467–474. Insulin blocks the stimulatory high-glucose effect on L-arginine transport by reducing transcriptional activity of the amino acid transporter through transcription factors specificity protein 1 and nuclear factor-κB. This may contribute to fetal endothelial dysfunction in diabetic pregnancies.
•• Lappas M, Hiden U, Froehlich J et al (2011) The Role of Oxidative Stress in the Pathophysiology of Gestational Diabetes Mellitus. Antioxid Redox Signal. This is a comprehensive review on pathogenesis of gestational diabetes and the oxidative and nitrative processes occurring in the placenta in this pathology.
Coughlan MT, Vervaart PP, Permezel M, et al. Altered placental oxidative stress status in gestational diabetes mellitus. Placenta. 2004;25:78–84.
Kwek K, Read MA, Khong TY, et al. Vasoactive effects of 8-epi-prostaglandin F(2alpha)in isolated human placental conduit and resistance blood vessels in vitro. Placenta. 2001;22:526–33.
Lappas M, Mitton A, Permezel M. In response to oxidative stress, the expression of inflammatory cytokines and antioxidant enzymes are impaired in placenta, but not adipose tissue, of women with gestational diabetes. J Endocrinol. 2010;204:75–84.
Singh U, Devaraj S, Jialal I. Vitamin E, oxidative stress, and inflammation. Annu Rev Nutr. 2005;25:151–74.
Bowen JM, Chamley L, Mitchell MD, et al. Cytokines of the placenta and extra-placental membranes: biosynthesis, secretion and roles in establishment of pregnancy in women. Placenta. 2002;23:239–56.
Marseille-Tremblay C, Ethier-Chiasson M, Forest JC, et al. Impact of maternal circulating cholesterol and gestational diabetes mellitus on lipid metabolism in human term placenta. Mol Reprod Dev. 2008;75:1054–62.
Coughlan MT, Oliva K, Georgiou HM, et al. Glucose-induced release of tumour necrosis factor-alpha from human placental and adipose tissues in gestational diabetes mellitus. Diabet Med. 2001;18:921–7.
Challier JC, Basu S, Bintein T, et al. Obesity in pregnancy stimulates macrophage accumulation and inflammation in the placenta. Placenta. 2008;29:274–81.
Lager S, Jansson N, Olsson AL, et al. Effect of IL-6 and TNF-alpha on fatty acid uptake in cultured human primary trophoblast cells. Placenta. 2011;32:121–7.
Magnusson AL, Waterman IJ, Wennergren M, et al. Triglyceride hydrolase activities and expression of fatty acid binding proteins in the human placenta in pregnancies complicated by intrauterine growth restriction and diabetes. J Clin Endocrinol Metab. 2004;89:4607–14.
• Gauster M, Hiden U, vanPoppel M et al (2011) Dysregulation of placental endothelial lipase in obese women with gestational diabetes mellitus. Diabetes in press. This demonstrates that neither GDM nor obesity alone but only their combination induces changes in a placental key lipase.
Gauster M, Hiden U, Blaschitz A, et al. Dysregulation of placental endothelial lipase and lipoprotein lipase in intrauterine growth-restricted pregnancies. J Clin Endocrinol Metab. 2007;92:2256–63.
Varastehpour A, Radaelli T, Minium J, et al. Activation of phospholipase A2 is associated with generation of placental lipid signals and fetal obesity. J Clin Endocrinol Metab. 2006;91:248–55.
•• Radaelli T, Lepercq J, Varastehpour A et al (2009) Differential regulation of genes for fetoplacental lipid pathways in pregnancy with gestational and type 1 diabetes mellitus. Am J Obstet Gynecol 201: 209 e201-209 e210. This demonstrates that GDM is a distinct diabetic entity in its effects on the placenta.
Jansson T, Ekstrand Y, Bjorn C, et al. Alterations in the activity of placental amino acid transporters in pregnancies complicated by diabetes. Diabetes. 2002;51:2214–9.
Kuruvilla AG, D'Souza SW, Glazier JD, et al. Altered activity of the system A amino acid transporter in microvillous membrane vesicles from placentas of macrosomic babies born to diabetic women. J Clin Invest. 1994;94:689–95.
Kalhan S, Parimi P. Gluconeogenesis in the fetus and neonate. Semin Perinatol. 2000;24:94–106.
Hahn T, Barth S, Weiss U, et al. Sustained hyperglycemia in vitro down-regulates the GLUT1 glucose transport system of cultured human term placental trophoblast: a mechanism to protect fetal development? Faseb J. 1998;12:1221–31.
Illsley NP, Hall S, Stacey TE. The modulation of glucose transfer across the human placenta by intervillous flow rates: An in vitro perfusion study. Troph Res. 1987;2:535–44.
Osmond DT, King RG, Brennecke SP, et al. Placental glucose transport and utilisation is altered at term in insulin-treated, gestational-diabetic patients. Diabetologia. 2001;44:1133–9.
Osmond DT, Nolan CJ, King RG, et al. Effects of gestational diabetes on human placental glucose uptake, transfer, and utilisation. Diabetologia. 2000;43:576–82.
• Colomiere M, Permezel M, Riley C et al (2009) Defective insulin signaling in placenta from pregnancies complicated by gestational diabetes mellitus. Eur J Endocrinol 160: 567–578. This compares the effects of GDM with and without obesity on insulin signaling in the placenta and shows varying effects, thus demonstrating the complexity of GDM-associated changes.
Disclosure
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gauster, M., Desoye, G., Tötsch, M. et al. The Placenta and Gestational Diabetes Mellitus. Curr Diab Rep 12, 16–23 (2012). https://doi.org/10.1007/s11892-011-0244-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11892-011-0244-5