Abstract
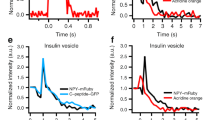
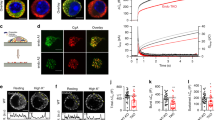
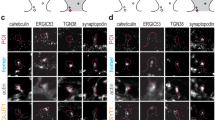
Synaptic terminals and neuroendocrine cells are packed with secretory vesicles, only a few of which are docked at the plasma membrane and readily releasable. The remainder are thought to constitute a large cytoplasmic reserve pool awaiting recruitment into the readily releasable pool (RRP) for exocytosis1. How vesicles are prioritized in recruitment is still unknown: the choice could be random, or else the oldest or the newest ones might be favoured. Here we show, using a fluorescent cargo protein2 that changes colour with time3, that vesicles in bovine adrenal chromaffin cells segregate into distinct populations, based on age. Newly assembled vesicles are immobile (morphologically docked) at the plasma membrane shortly after biogenesis, whereas older vesicles are mobile and located deeper in the cell. Different secretagogues selectively release vesicles from the RRP or, surprisingly, selectively from the deeper cytoplasmic pool. Thus, far from being equal, vesicles are segregated functionally and spatially according to age.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Morgan, A. & Burgoyne, R. D. in Seminars in Cell and Developmental Biology (eds Burgoyne, R. D. & Mombaerts, P.) 141–149 (Academic, Cambridge, 1997)
Burke, N. V. et al. Neuronal peptide release is limited by secretory granule mobility. Neuron 19, 1095–1102 (1997)
Terskikh, A. et al. ‘Fluorescent timer’: protein that changes colour with time. Science 290, 1585–1588 (2000)
Nguyen, T. T., Ong, H. & De Lean, A. Secretion and biosynthesis of atrial natriuretic factor by cultured adrenal chromaffin cells. FEBS Lett. 231, 393–396 (1988)
Cuchillo-Ibanez, I., Michelena, P., Albillos, A. & Garcia, A. G. A preferential pole for exocytosis in cultured chromaffin cells revealed by confocal microscopy. FEBS Lett. 459, 22–26 (1999)
Phillips, J. H. Dynamic aspects of chromaffin granule structure. Neuroscience 7, 1595–1609 (1982)
Corcoran, J. J., Wilson, S. P. & Kirshner, N. Flux of catecholamines through chromaffin vesicles in cultured bovine adrenal medullary cells. J. Biol. Chem. 259, 6208–6214 (1984)
Burgoyne, R. D. Control of exocytosis in adrenal chromaffin cells. Biochim. Biophys. Acta 1071, 174–202 (1991)
Neher, E. & Zucker, R. S. Multiple calcium-dependent prosesses related to secretion in bovine chromaffin cells. Neuron 10, 21–30 (1993)
Parsons, T. D., Coorssen, J. R., Horstmann, H. & Almers, W. Docked granules, the exocytic burst, and the need for ATP hydrolysis in endocrine cells. Neuron 15, 1085–1096 (1995)
Steyer, J. A., Horstmann, H. & Almers, W. Transport, docking and exocytosis of single secretory granules in live chromaffin cells. Nature 388, 474–478 (1997)
Oheim, M., Loerke, D., Stuhmer, W. & Chow, R. H. The last few milliseconds in the life of a secretory granule. Docking, dynamics and fusion visualized by total internal reflection fluorescence microscopy (TIRFM). Eur. Biophys. J. 27, 83–98 (1998)
Seward, E. P., Chernevskaya, N. I. & Nowycky, M. C. Ba2+ ions evoke two kinetically distinct patterns of exocytosis in chromaffin cells, but not in neurohypophysial nerve terminals. J. Neurosci. 16, 1370–1379 (1996)
Neves, G., Neef, A. & Lagnado, L. The actions of barium and strontium on exocytosis and endocytosis in the synaptic terminal of goldfish bipolar cells. J. Physiol. (Lond.) 535, 809–824 (2001)
TerBush, D. R. & Holz, R. W. Barium and calcium stimulate secretion from digitonin-permeabilized bovine adrenal chromaffin cells by similar pathways. J. Neurochem. 58, 680–687 (1992)
Heldman, E., Levine, M., Raveh, L. & Pollard, H. B. Barium ions enter chromaffin cells via voltage-dependent calcium channels and induce secretion by a mechanism independent of calcium. J. Biol. Chem. 264, 7914–7920 (1989)
Han, W., Ng, Y. K., Axelrod, D. & Levitan, E. S. Neuropeptide release by efficient recruitment of diffusing cytoplasmic secretory vesicles. Proc. Natl Acad. Sci. USA 96, 14577–14582 (1999)
Ogawa, T., Vatta, M., Bruneau, B. G. & de Bold, A. J. Characterization of natriuretic peptide production by adult heart atria. Am. J. Physiol. 276, H1977–H1986 (1999)
Chen, T. T. et al. Relative importance of newly synthesized and stored hormone to basal secretion by growth hormone and prolactin cells. Endocrinology 125, 1904–1909 (1989)
Rudolf, R., Salm, T., Rustom, A. & Gerdes, H. H. Dynamics of immature secretory granules: role of cytoskeletal elements during transport, cortical restriction, and F-actin-dependent tethering. Mol. Biol. Cell 12, 1353–1365 (2001)
Sugita, S. et al. Synaptotagmins form a hierarchy of exocytotic Ca2+ sensors with distinct Ca2+ affinities. EMBO J. 21, 270–280 (2002)
Li, C. et al. Ca2+-dependent and Ca2+-independent activities of neural and nonneural synaptotagmins. Nature 375, 594–599 (1995)
Winkler, H. & Fischer-Colbrie, R. Regulation of the biosynthesis of large dense-core vesicles in chromaffin cells and neurons. Cell. Mol. Neurobiol. 18, 193–209 (1998)
Lundstrom, K. et al. Novel mutant Semliki Forest virus vectors: gene expression and localization studies in neuronal cells. Histochem. Cell. Biol. 115, 83–91 (2001)
Duncan, R. R. et al. High-efficiency Semliki Forest virus-mediated transduction in bovine adrenal chromaffin cells. Biochem. J. 342, 497–501 (1999)
Baird, G. S., Zacharias, D. A. & Tsien, R. Y. Biochemistry, mutagenesis, and oligomerization of DsRed, a red fluorescent protein from coral. Proc. Natl Acad. Sci. USA 97, 11984–11989 (2000)
Freestone, S. et al. The renal effects of atrial natriuretic peptide in man are not attenuated by (+ )-sulpiride. Br. J. Clin. Pharmacol. 27, 13–18 (1989)
Acknowledgements
We thank A. Morgan for the catecholamine assay protocol; J. Pryde for the polyclonal anti-Golgi serum; D. J. Webb and N. Johnston for the anti-ANF serum and RIA protocol; and L. Sharp for assistance with confocal microscopy. This work was funded by Wellcome Trust grants and an NIH grant to D.K.A., R.H.C. and M.J.S.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 246 kb)
Rights and permissions
About this article
Cite this article
Duncan, R., Greaves, J., Wiegand, U. et al. Functional and spatial segregation of secretory vesicle pools according to vesicle age. Nature 422, 176–180 (2003). https://doi.org/10.1038/nature01389
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01389
This article is cited by
-
Neuroendocrine neoplasia of the gastrointestinal tract revisited: towards precision medicine
Nature Reviews Endocrinology (2020)
-
Syntaxin-4 mediates exocytosis of pre-docked and newcomer insulin granules underlying biphasic glucose-stimulated insulin secretion in human pancreatic beta cells
Diabetologia (2015)
-
Cholesterol effects on vesicle pools in chromaffin cells revealed by carbon-fiber microelectrode amperometry
Analytical and Bioanalytical Chemistry (2011)
-
Repetitive Stimulation of the Pituitary with Growth-Hormone-Releasing Hormone Alters the Proportion of 22 and 20 Kilodalton Human-Growth Hormone Released
International Journal of Pediatric Endocrinology (2010)
-
Monomeric fluorescent timers that change color from blue to red report on cellular trafficking
Nature Chemical Biology (2009)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.