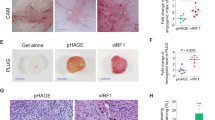
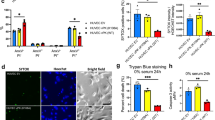
Abstract
Kaposi Sarcoma (KS) are opportunistic tumors, associated with human herpes virus 8 (HHV8) infection. KS development is highly favored by immune-depression and remains the second most frequent tumor in acquired immune deficiency syndrome patients. Although it has been shown that experimental expression of the HHV8 G-protein-coupled receptor (vGPCR) in the endothelial compartment is alone sufficient to recapitulate the formation and progression of KS-like lesions, its functional effects on endothelial homeostasis are not fully understood. Here we show that vGPCR expression in endothelial cells induces an increase in paracellular permeability both in vivo and in vitro. By using pharmacological inhibitors and small interference RNA-based knockdown, we demonstrate an essential role for the PI(3)Kinase-γ/Rac nexus in vGPCR-mediated permeability. This was further accompanied by dramatic remodeling of VE-cadherin-dependent cell–cell junctions. Importantly, this in vitro vGPCR-initiated signaling signature was observed in a large panel of human KS. Altogether, our results support the hypothesis that endothelial vGPCR signaling is co-opted in KS, and unveil new key cellular targets for therapeutic intervention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ascherl G, Hohenadl C, Schatz O, Shumay E, Bogner J, Eckhart L et al (1999). Infection with human immunodeficiency virus-1 increases expression of vascular endothelial cell growth factor in T cells: implications for acquired immunodeficiency syndrome-associated vasculopathy. Blood 93: 4232–4241.
Bais C, Santomasso B, Coso O, Arvanitakis L, Raaka EG, Gutkind JS et al. (1998). G-protein-coupled receptor of Kaposi′s sarcoma-associated herpesvirus is a viral oncogene and angiogenesis activator. Nature 391: 86–89.
Barber DF, Bartolome A, Hernandez C, Flores JM, Redondo C, Fernandez-Arias C et al. (2005). PI3Kgamma inhibition blocks glomerulonephritis and extends lifespan in a mouse model of systemic lupus. Nat Med 11: 933–935.
Camps M, Ruckle T, Ji H, Ardissone V, Rintelen F, Shaw J et al. (2005). Blockade of PI3Kgamma suppresses joint inflammation and damage in mouse models of rheumatoid arthritis. Nat Med 11: 936–943.
Cesarman E, Mesri EA . (2007). Kaposi sarcoma-associated herpesvirus and other viruses in human lymphomagenesis. Curr Top Microbiol Immunol 312: 263–287.
Chaisuparat R, Hu J, Jham BC, Knight ZA, Shokat KM, Montaner S . (2008). Dual inhibition of PI3Kalpha and mTOR as an alternative treatment for Kaposi's sarcoma. Cancer Res 68: 8361–8368.
Chang Y, Cesarman E, Pessin MS, Lee F, Culpepper J, Knowles DM et al. (1994). Identification of herpesvirus-like DNA sequences in AIDS-associated Kaposi's sarcoma. Science 266: 1865–1869.
Chiou CJ, Poole LJ, Kim PS, Ciufo DM, Cannon JS, ap Rhys CM et al. (2002). Patterns of gene expression and a transactivation function exhibited by the vGCR (ORF74) chemokine receptor protein of Kaposi's sarcoma-associated herpesvirus. J Virol 76: 3421–3439.
Dejana E . (2004). Endothelial cell-cell junctions: happy together. Nat Rev Mol Cell Biol 5: 261–270.
Dupin N, Fisher C, Kellam P, Ariad S, Tulliez M, Franck N et al. (1999). Distribution of human herpesvirus-8 latently infected cells in Kaposi′s sarcoma, multicentric Castleman′s disease, and primary effusion lymphoma. Proc Natl Acad Sci USA 96: 4546–4551.
Dupin N, Grange PA . (2006). Looking for the target cell of Kaposi's sarcoma-associated herpesvirus. J Invest Dermatol 126: 545–547.
Gavard J, Gutkind JS . (2006). VEGF controls endothelial-cell permeability by promoting the beta-arrestin-dependent endocytosis of VE-cadherin. Nat Cell Biol 8: 1223–1234.
Gavard J, Hou X, Qu Y, Masedunskas A, Martin D, Weigert R et al. (2009). A role for a CXCR2/phosphatidylinositol 3-kinase gamma signaling axis in acute and chronic vascular permeability. Mol Cell Biol 29: 2469–2480.
Gavard J, Lambert M, Grosheva I, Marthiens V, Irinopoulou T, Riou JF et al. (2004). Lamellipodium extension and cadherin adhesion: two cell responses to cadherin activation relying on distinct signalling pathways. J Cell Sci 117: 257–270.
Gavard J, Patel V, Gutkind JS . (2008). Angiopoietin-1 prevents VEGF-induced endothelial permeability by sequestering Src through mDia. Dev Cell 14: 25–36.
Grisotto MG, Garin A, Martin AP, Jensen KK, Chan P, Sealfon SC et al. (2006). The human herpesvirus 8 chemokine receptor vGPCR triggers autonomous proliferation of endothelial cells. J Clin Invest 116: 1264–1273.
Grundhoff A, Ganem D . (2004). Inefficient establishment of KSHV latency suggests an additional role for continued lytic replication in Kaposi sarcoma pathogenesis. J Clin Invest 113: 124–136.
Martin D, Galisteo R, Ji Y, Montaner S, Gutkind JS . (2008). An NF-kappaB gene expression signature contributes to Kaposi′s sarcoma virus vGPCR-induced direct and paracrine neoplasia. Oncogene 27: 1844–1852.
Maurer T, Ponte M, Leslie K . (2007). HIV-associated Kaposi's sarcoma with a high CD4 count and a low viral load. N Engl J Med 357: 1352–1353.
Mitsuyasu RT . (2000). Update on the pathogenesis and treatment of Kaposi sarcoma. Curr Opin Oncol 12: 174–180.
Monini P, Sgadari C, Grosso MG, Bellino S, Di Biagio A, Toschi E et al. (2009). Clinical course of classic Kaposi's sarcoma in HIV-negative patients treated with the HIV protease inhibitor indinavir. AIDS 23: 534–538.
Montaner S, Sodhi A, Molinolo A, Bugge TH, Sawai ET, He Y et al. (2003). Endothelial infection with KSHV genes in vivo reveals that vGPCR initiates Kaposi's sarcomagenesis and can promote the tumorigenic potential of viral latent genes. Cancer Cell 3: 23–36.
Montaner S, Sodhi A, Pece S, Mesri EA, Gutkind JS . (2001). The Kaposi's sarcoma-associated herpesvirus G protein-coupled receptor promotes endothelial cell survival through the activation of Akt/protein kinase B. Cancer Res 61: 2641–2648.
Montaner S, Sodhi A, Ramsdell AK, Martin D, Hu J, Sawai ET et al. (2006). The Kaposi's sarcoma-associated herpesvirus G protein-coupled receptor as a therapeutic target for the treatment of Kaposi's sarcoma. Cancer Res 66: 168–174.
Montaner S, Sodhi A, Servitja JM, Ramsdell AK, Barac A, Sawai ET et al. (2004). The small GTPase Rac1 links the Kaposi sarcoma-associated herpesvirus vGPCR to cytokine secretion and paracrine neoplasia. Blood 104: 2903–2911.
Mutlu AD, Cavallin LE, Vincent L, Chiozzini C, Eroles P, Duran EM et al. (2007). In vivo-restricted and reversible malignancy induced by human herpesvirus-8 KSHV: a cell and animal model of virally induced Kaposi's sarcoma. Cancer Cell 11: 245–258.
Oksenhendler E, Boulanger E, Galicier L, Du MQ, Dupin N, Diss TC et al. (2002). High incidence of Kaposi sarcoma-associated herpesvirus-related non-Hodgkin lymphoma in patients with HIV infection and multicentric Castleman disease. Blood 99: 2331–2336.
Orenstein JM, Alkan S, Blauvelt A, Jeang KT, Weinstein MD, Ganem D et al. (1997). Visualization of human herpesvirus type 8 in Kaposi's sarcoma by light and transmission electron microscopy. AIDS 11: F35–F45.
Qian LW, Greene W, Ye F, Gao SJ . (2008). Kaposi's sarcoma-associated herpesvirus disrupts adherens junctions and increases endothelial permeability by inducing degradation of VE-cadherin. J Virol 82: 11902–11912.
Sgadari C, Barillari G, Toschi E, Carlei D, Bacigalupo I, Baccarini S et al. (2002). HIV protease inhibitors are potent anti-angiogenic molecules and promote regression of Kaposi sarcoma. Nat Med 8: 225–232.
Sodhi A, Chaisuparat R, Hu J, Ramsdell AK, Manning BD, Sausville EA et al. (2006). The TSC2/mTOR pathway drives endothelial cell transformation induced by the Kaposi's sarcoma-associated herpesvirus G protein-coupled receptor. Cancer Cell 10: 133–143.
Sodhi A, Montaner S, Gutkind JS . (2004a). Viral hijacking of G-protein-coupled-receptor signalling networks. Nat Rev Mol Cell Biol 5: 998–1012.
Sodhi A, Montaner S, Patel V, Gomez-Roman JJ, Li Y, Sausville EA et al. (2004b). Akt plays a central role in sarcomagenesis induced by Kaposi's sarcoma herpesvirus-encoded G protein-coupled receptor. Proc Natl Acad Sci USA 101: 4821–4826.
Sodhi A, Montaner S, Patel V, Zohar M, Bais C, Mesri EA et al. (2000). The Kaposi's sarcoma-associated herpes virus G protein-coupled receptor up-regulates vascular endothelial growth factor expression and secretion through mitogen-activated protein kinase and p38 pathways acting on hypoxia-inducible factor 1alpha. Cancer Res 60: 4873–4880.
Stallone G, Schena A, Infante B, Di Paolo S, Loverre A, Maggio G et al. (2005). Sirolimus for Kaposi's sarcoma in renal-transplant recipients. N Engl J Med 352: 1317–1323.
Szajerka T, Jablecki J . (2007). Kaposi's sarcoma revisited. AIDS Rev 9: 230–236.
Taddei A, Giampietro C, Conti A, Orsenigo F, Breviario F, Pirazzoli V et al. (2008). Endothelial adherens junctions control tight junctions by VE-cadherin-mediated upregulation of claudin-5. Nat Cell Biol 10: 923–934.
Weis SM, Cheresh DA . (2005). Pathophysiological consequences of VEGF-induced vascular permeability. Nature 437: 497–504.
Acknowledgements
We would like to thank N. Bidère (Inserm U1014, Université Paris-Sud, Hopital Paul Brousse, Villejuif, France) for helpful discussions and comments, A Schmitt (Institut Cochin, Paris, France) for technical help on electron microscopy experiments, Anne Audebourg and Brigitte Radenen (Service d'Anatomie et de Cytologie Pathologique, Hôpital Cochin, Paris, France) for technical help on histological staining, and Nina Feinberg (OPCB, NIDCR, NIH, Bethesda, MD, USA) for technical help on the initial project. This research was funded by Ligue Nationale contre le Cancer, comité de Paris and by a Marie Curie International Reintegration Grant within the 7th European Community Framework Programme (PIRG04-GA-2008-239126). JD is supported by a post-doctoral fellowship from Fondation pour la Recherche Médicale, ALG by the PhD program from Université Paris 5 and EMGM by a post-doctoral fellowship from Association pour la Recherche sur le Cancer.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Dwyer, J., Le Guelte, A., Galan Moya, E. et al. Remodeling of VE-cadherin junctions by the human herpes virus 8 G-protein coupled receptor. Oncogene 30, 190–200 (2011). https://doi.org/10.1038/onc.2010.411
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.411
Keywords
This article is cited by
-
The guanine exchange factor SWAP70 mediates vGPCR-induced endothelial plasticity
Cell Communication and Signaling (2015)
-
Recombinant Tissue-Type Plasminogen Activator Transiently Enhances Blood–Brain Barrier Permeability During Cerebral Ischemia through Vascular Endothelial Growth Factor-Mediated Endothelial Endocytosis in Mice
Journal of Cerebral Blood Flow & Metabolism (2015)
-
YGLF motif in the Kaposi sarcoma herpes virus G-protein-coupled receptor adjusts NF-κB activation and paracrine actions
Oncogene (2014)
-
Human tumour viruses and the deregulation of cell polarity in cancer
Nature Reviews Cancer (2012)