Abstract
The prevalence of obesity in combination with sarcopenia (the age-related loss of muscle mass and strength or physical function) is increasing in adults aged 65 years and older. A major subset of adults over the age of 65 is now classified as having sarcopenic obesity, a high-risk geriatric syndrome predominantly observed in an ageing population that is at risk of synergistic complications from both sarcopenia and obesity. This Review discusses pathways and mechanisms leading to muscle impairment in older adults with obesity. We explore sex-specific hormonal changes, inflammatory pathways and myocellular mechanisms leading to the development of sarcopenic obesity. We discuss the evolution, controversies and challenges in defining sarcopenic obesity and present current body composition modalities used to assess this condition. Epidemiological surveys form the basis of defining its prevalence and consequences beyond comorbidity and mortality. Current treatment strategies, and the evidence supporting them, are outlined, with a focus on calorie restriction, protein supplementation and aerobic and resistance exercises. We also describe weight loss-induced complications in patients with sarcopenic obesity that are relevant to clinical management. Finally, we review novel and potential future therapies including testosterone, selective androgen receptor modulators, myostatin inhibitors, ghrelin analogues, vitamin K and mesenchymal stem cell therapy.
Key points
-
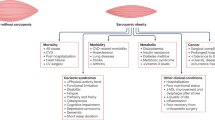
Body composition changes that occur with the ageing process can lead to sarcopenic obesity, an increasingly prevalent disorder owing to the increased prevalence of obesity in an ageing population.
-
Hormonal, inflammatory and myocellular mechanisms impact underlying biological processes that promote fat deposition and loss of lean mass and strength.
-
Definitions of sarcopenia and obesity can vary considerably, prompting difficulties in the diagnosis and epidemiological understanding of sarcopenic obesity as well as the development of treatment strategies for this disease.
-
Lifestyle interventions including calorie restriction and physical activity consisting of aerobic and resistance exercises are the cornerstones of therapy.
-
Clinicians and researchers need to be aware of weight loss-induced sarcopenia and osteopenia.
-
Novel, promising therapies, including weight loss medications, bariatric surgery, whole-body vibration therapy, periodization (a systematic variation in physical training specificity, intensity and volume within periods), testosterone, selective androgen receptor modulators, anamorelin, myostatin inhibitors, vitamin K and mesenchymal stem cells, require further investigation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
United Nations, Department of Economic and Social Affairs, Population Division. World Population Prospects: The 2017 Revision, Key Findings and Advance Tables. Working Paper No. ESA/P/WP/248 (United Nations, 2017).
Flegal, K. M., Kruszon-Moran, D., Carroll, M. D., Fryar, C. D. & Ogden, C. L. Trends in obesity among adults in the United States, 2005 to 2014. JAMA 315, 2284–2291 (2016).
Johnson, N. B., Hayes, L. D., Brown, K., Hoo, E. C. & Ethier, K. A. CDC National Health Report: leading causes of morbidity and mortality and associated behavioral risk and protective factors—United States, 2005–2013. MMWR Suppl. 63, 3–27 (2014).
Hales, C. M., Carroll, M. D., Fryar, C. D. & Ogden, C. L. Prevalence of obesity among adults and youth: United States, 2015–2016. Centers for Disease Control and Prevention https://www.cdc.gov/nchs/data/databriefs/db288.pdf (2017). These are the most updated prevalence rates of the obesity epidemic in the United States.
Organisation for Economic Co-operation and Development. Obesity Update 2017. OECD www.oecd.org/health/obesity-update.htm (2017).
Acciai, F. & Firebaugh, G. Why did life expectancy decline in the United States in 2015? A gender-specific analysis. Soc. Sci. Med. 190, 174–180 (2017).
Ludwig, D. S. Lifespan weighed down by diet. JAMA 315, 2269–2270 (2016).
Olshansky, S. J. et al. A potential decline in life expectancy in the United States in the 21st century. N. Engl. J. Med. 352, 1138–1145 (2005).
Preston, S. H., Stokes, A., Mehta, N. K. & Cao, B. Projecting the effect of changes in smoking and obesity on future life expectancy in the United States. Demography 51, 27–49 (2014).
Stewart, S. T., Cutler, D. M. & Rosen, A. B. Forecasting the effects of obesity and smoking on U.S. life expectancy. N. Engl. J. Med. 361, 2252–2260 (2009).
Roubenoff, R. Sarcopenic obesity: the confluence of two epidemics. Obes. Res. 12, 887–888 (2004).
Patterson, R. E., Frank, L. L., Kristal, A. R. & White, E. A comprehensive examination of health conditions associated with obesity in older adults. Am. J. Prev. Med. 27, 385–390 (2004).
Flegal, K. M. et al. Comparisons of percentage body fat, body mass index, waist circumference, and waist-stature ratio in adults. Am. J. Clin. Nutr. 89, 500–508 (2009).
Heo, M., Faith, M. S., Pietrobelli, A. & Heymsfield, S. B. Percentage of body fat cutoffs by sex, age, and race-ethnicity in the US adult population from NHANES 1999–2004. Am. J. Clin. Nutr. 95, 594–602 (2012).
Xu, W. et al. Height loss, vertebral fractures, and the misclassification of osteoporosis. Bone 48, 307–311 (2011).
Sayer, A. A. et al. The developmental origins of sarcopenia. J. Nutr. Health Aging 12, 427–432 (2008). This is a review that evaluates the development of and life-course influences on muscle mass and strength.
Abizanda, P. et al. Energetics of aging and frailty: the FRADEA Study. J. Gerontol. A Biol. Sci. Med. Sci. 71, 787–796 (2016).
Weinsier, R. L., Schutz, Y. & Bracco, D. Reexamination of the relationship of resting metabolic rate to fat-free mass and to the metabolically active components of fat-free mass in humans. Am. J. Clin. Nutr. 55, 790–794 (1992).
Gallagher, D. et al. Organ-tissue mass measurement allows modeling of REE and metabolically active tissue mass. Am. J. Physiol. 275, E249–E258 (1998).
Wilson, M. M. & Morley, J. E. Invited review: aging and energy balance. J. Appl. Physiol. (1985) 95, 1728–1736 (2003).
Conley, K. E. et al. Ageing, muscle properties and maximal O(2) uptake rate in humans. J. Physiol. 526, 211–217 (2000).
Conley, K. E., Jubrias, S. A. & Esselman, P. C. Oxidative capacity and ageing in human muscle. J. Physiol. 526, 203–210 (2000).
Cannon, B. & Nedergaard, J. Nonshivering thermogenesis and its adequate measurement in metabolic studies. J. Exp. Biol. 214, 242–253 (2011).
Dulloo, A. G., Seydoux, J. & Jacquet, J. Adaptive thermogenesis and uncoupling proteins: a reappraisal of their roles in fat metabolism and energy balance. Physiol. Behav. 83, 587–602 (2004).
Doucet, E. et al. Appetite after weight loss by energy restriction and a low-fat diet-exercise follow-up. Int. J. Obes. Relat. Metab. Disord. 24, 906–914 (2000).
Doucet, E. et al. Changes in energy expenditure and substrate oxidation resulting from weight loss in obese men and women: is there an important contribution of leptin? J. Clin. Endocrinol. Metab. 85, 1550–1556 (2000).
Bray, G. A. Effect of caloric restriction on energy expenditure in obese patients. Lancet 2, 397–398 (1969). This is an early article that highlights the metabolic changes with energy restriction in patients with obesity.
Major, G. C., Doucet, E., Trayhurn, P., Astrup, A. & Tremblay, A. Clinical significance of adaptive thermogenesis. Int. J. Obes. (Lond.) 31, 204–212 (2007).
Cohn, S. H. et al. Changes in body chemical composition with age measured by total-body neutron activation. Metabolism 25, 85–95 (1976).
Tremblay, A., Royer, M. M., Chaput, J. P. & Doucet, E. Adaptive thermogenesis can make a difference in the ability of obese individuals to lose body weight. Int. J. Obes. (Lond.) 37, 759–764 (2013).
Astrup, A. et al. Meta-analysis of resting metabolic rate in formerly obese subjects. Am. J. Clin. Nutr. 69, 1117–1122 (1999).
Rosenbaum, M., Hirsch, J., Gallagher, D. A. & Leibel, R. L. Long-term persistence of adaptive thermogenesis in subjects who have maintained a reduced body weight. Am. J. Clin. Nutr. 88, 906–912 (2008).
Napier, J. R., Thomas, M. F., Sharma, M., Hodgkinson, S. C. & Bass, J. J. Insulin-like growth factor-I protects myoblasts from apoptosis but requires other factors to stimulate proliferation. J. Endocrinol. 163, 63–68 (1999).
Roth, S. M. et al. Ultrastructural muscle damage in young versus older men after high-volume, heavy-resistance strength training. J. Appl. Physiol. (1985) 86, 1833–1840 (1999).
Klem, M. L., Wing, R. R., Lang, W., McGuire, M. T. & Hill, J. O. Does weight loss maintenance become easier over time? Obes. Res. 8, 438–444 (2000).
Wing, R. R. & Hill, J. O. Successful weight loss maintenance. Annu. Rev. Nutr. 21, 323–341 (2001).
Tremollieres, F. A., Pouilles, J. M. & Ribot, C. A. Relative influence of age and menopause on total and regional body composition changes in postmenopausal women. Am. J. Obstet. Gynecol. 175, 1594–1600 (1996).
Sowers, M. et al. Changes in body composition in women over six years at midlife: ovarian and chronological aging. J. Clin. Endocrinol. Metab. 92, 895–901 (2007).
Milewicz, A., Tworowska, U. & Demissie, M. Menopausal obesity — myth or fact? Climacteric 4, 273–283 (2001).
Gambacciani, M. et al. Climacteric modifications in body weight and fat tissue distribution. Climacteric 2, 37–44 (1999).
Guo, S. S., Zeller, C., Chumlea, W. C. & Siervogel, R. M. Aging, body composition, and lifestyle: the Fels Longitudinal Study. Am. J. Clin. Nutr. 70, 405–411 (1999). This study presents the natural patterns of change in body composition in older adults and the influence of physical activity.
Enns, D. L. & Tiidus, P. M. Estrogen influences satellite cell activation and proliferation following downhill running in rats. J. Appl. Physiol. (1985) 104, 347–353 (2008).
Gambacciani, M. et al. Prospective evaluation of body weight and body fat distribution in early postmenopausal women with and without hormonal replacement therapy. Maturitas 39, 125–132 (2001).
Kadi, F. Cellular and molecular mechanisms responsible for the action of testosterone on human skeletal muscle. A basis for illegal performance enhancement. Br. J. Pharmacol. 154, 522–528 (2008).
Bondanelli, M. et al. Activation of the somatotropic axis by testosterone in adult men: evidence for a role of hypothalamic growth hormone-releasing hormone. Neuroendocrinology 77, 380–387 (2003).
Yeap, B. B. Are declining testosterone levels a major risk factor for ill-health in aging men? Int. J. Impot. Res. 21, 24–36 (2009).
LeBlanc, E. S. et al. Higher testosterone levels are associated with less loss of lean body mass in older men. J. Clin. Endocrinol. Metab. 96, 3855–3863 (2011).
Miller, K. K. Androgen deficiency: effects on body composition. Pituitary 12, 116–124 (2009).
Kaplan, S. A., Lee, J. Y., O’Neill, E. A., Meehan, A. G. & Kusek, J. W. Prevalence of low testosterone and its relationship to body mass index in older men with lower urinary tract symptoms associated with benign prostatic hyperplasia. Aging Male 16, 169–172 (2013).
Urban, R. J. et al. Testosterone administration to elderly men increases skeletal muscle strength and protein synthesis. Am. J. Physiol. 269, E820–E826 (1995).
Snyder, P. J. et al. Effect of testosterone treatment on body composition and muscle strength in men over 65 years of age. J. Clin. Endocrinol. Metab. 84, 2647–2653 (1999).
Ferrando, A. A. et al. Testosterone administration to older men improves muscle function: molecular and physiological mechanisms. Am. J. Physiol. Endocrinol. Metab. 282, E601–E607 (2002).
Snyder, P. J. et al. Effects of testosterone treatment in older men. N. Engl. J. Med. 374, 611–624 (2016). This is a randomized trial of testosterone therapy in symptomatic older men that demonstrates no benefit in vitality or walking distance following supplementation.
Orentreich, N., Brind, J. L., Rizer, R. L. & Vogelman, J. H. Age changes and sex differences in serum dehydroepiandrosterone sulfate concentrations throughout adulthood. J. Clin. Endocrinol. Metab. 59, 551–555 (1984).
Nafziger, A. N., Bowlin, S. J., Jenkins, P. L. & Pearson, T. A. Longitudinal changes in dehydroepiandrosterone concentrations in men and women. J. Lab. Clin. Med. 131, 316–323 (1998).
Giannoulis, M. G., Martin, F. C., Nair, K. S., Umpleby, A. M. & Sonksen, P. Hormone replacement therapy and physical function in healthy older men. Time to talk hormones? Endocr. Rev. 33, 314–377 (2012).
Schrager, M. A. et al. Sarcopenic obesity and inflammation in the InCHIANTI study. J. Appl. Physiol. (1985) 102, 919–925 (2007). This is an epidemiological study that shows that obesity directly affects inflammation and negatively affects muscle strength.
Forsythe, L. K., Wallace, J. M. & Livingstone, M. B. Obesity and inflammation: the effects of weight loss. Nutr. Res. Rev. 21, 117–133 (2008).
Park, H. S., Park, J. Y. & Yu, R. Relationship of obesity and visceral adiposity with serum concentrations of CRP, TNF-alpha and IL-6. Diabetes Res. Clin. Pract. 69, 29–35 (2005).
Zamboni, M., Mazzali, G., Fantin, F., Rossi, A. & Di Francesco, V. Sarcopenic obesity: a new category of obesity in the elderly. Nutr. Metab. Cardiovasc. Dis. 18, 388–395 (2008).
Hamrick, M. W. Role of the cytokine-like hormone leptin in muscle-bone crosstalk with aging. J. Bone Metab. 24, 1–8 (2017).
Yeap, B. B. et al. Associations of insulin-like growth factor-I and its binding proteins and testosterone with frailty in older men. Clin. Endocrinol. (Oxf.) 78, 752–759 (2013).
Wang, Y. et al. Adiponectin inhibits tumor necrosis factor-alpha-induced vascular inflammatory response via caveolin-mediated ceramidase recruitment and activation. Circ. Res. 114, 792–805 (2014).
Lang, C. H., Frost, R. A., Nairn, A. C., MacLean, D. A. & Vary, T. C. TNF-alpha impairs heart and skeletal muscle protein synthesis by altering translation initiation. Am. J. Physiol. Endocrinol. Metab. 282, E336–E347 (2002).
Cartwright, M. J., Tchkonia, T. & Kirkland, J. L. Aging in adipocytes: potential impact of inherent, depot-specific mechanisms. Exp. Gerontol. 42, 463–471 (2007).
Shulman, G. I. Ectopic fat in insulin resistance, dyslipidemia, and cardiometabolic disease. N. Engl. J. Med. 371, 1131–1141 (2014).
Thomas, D. R. Loss of skeletal muscle mass in aging: examining the relationship of starvation, sarcopenia and cachexia. Clin. Nutr. 26, 389–399 (2007).
Nilwik, R. et al. The decline in skeletal muscle mass with aging is mainly attributed to a reduction in type II muscle fiber size. Exp. Gerontol. 48, 492–498 (2013).
Delbono, O. Neural control of aging skeletal muscle. Aging Cell 2, 21–29 (2003).
Stenholm, S. et al. Sarcopenic obesity: definition, cause and consequences. Curr. Opin. Clin. Nutr. Metab. Care 11, 693–700 (2008).
Bauer, J. et al. Evidence-based recommendations for optimal dietary protein intake in older people: a position paper from the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 14, 542–559 (2013). This is a position paper from the PROT-Age study group that recommends an average daily intake of at least 1.0–1.2 g protein per kg body weight per day.
Gallagher, D. et al. Healthy percentage body fat ranges: an approach for developing guidelines based on body mass index. Am. J. Clin. Nutr. 72, 694–701 (2000). This study includes ethnically heterogeneous study populations, which allow the development of prediction formulas for healthy percentage body fat.
Goodpaster, B. H. et al. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J. Gerontol. A Biol. Sci. Med. Sci. 61, 1059–1064 (2006). This study shows that loss of strength with ageing is much more rapid than the concomitant loss of muscle mass, suggesting a decline in muscle quality.
Visser, M. et al. Muscle mass, muscle strength, and muscle fat infiltration as predictors of incident mobility limitations in well-functioning older persons. J. Gerontol. A Biol. Sci. Med. Sci. 60, 324–333 (2005). This study shows that in sarcopenic obesity reflected by low cross-sectional thigh muscle area, greater fat infiltration into muscle is associated with mobility loss.
Sepe, A., Tchkonia, T., Thomou, T., Zamboni, M. & Kirkland, J. L. Aging and regional differences in fat cell progenitors — a mini-review. Gerontology 57, 66–75 (2011).
Kalinkovich, A. & Livshits, G. Sarcopenic obesity or obese sarcopenia: a cross talk between age-associated adipose tissue and skeletal muscle inflammation as a main mechanism of the pathogenesis. Ageing Res. Rev. 35, 200–221 (2017).
Kob, R. et al. Sarcopenic obesity: molecular clues to a better understanding of its pathogenesis? Biogerontology 16, 15–29 (2015).
Stinkens, R., Goossens, G. H., Jocken, J. W. & Blaak, E. E. Targeting fatty acid metabolism to improve glucose metabolism. Obes. Rev. 16, 715–757 (2015).
Aon, M. A., Bhatt, N. & Cortassa, S. C. Mitochondrial and cellular mechanisms for managing lipid excess. Front. Physiol. 5, 282 (2014).
Bruce, C. R. et al. Muscle oxidative capacity is a better predictor of insulin sensitivity than lipid status. J. Clin. Endocrinol. Metab. 88, 5444–5451 (2003).
Kohara, K. Sarcopenic obesity in aging population: current status and future directions for research. Endocrine 45, 15–25 (2014).
Carnio, S. et al. Autophagy impairment in muscle induces neuromuscular junction degeneration and precocious aging. Cell Rep. 8, 1509–1521 (2014).
Marcell, T. J. Sarcopenia: causes, consequences, and preventions. J. Gerontol. A Biol. Sci. Med. Sci. 58, M911–M916 (2003).
Wohlgemuth, S. E., Seo, A. Y., Marzetti, E., Lees, H. A. & Leeuwenburgh, C. Skeletal muscle autophagy and apoptosis during aging: effects of calorie restriction and life-long exercise. Exp. Gerontol. 45, 138–148 (2010).
Manini, T. M. et al. Reduced physical activity increases intermuscular adipose tissue in healthy young adults. Am. J. Clin. Nutr. 85, 377–384 (2007).
Kusminski, C. M., Shetty, S., Orci, L., Unger, R. H. & Scherer, P. E. Diabetes and apoptosis: lipotoxicity. Apoptosis 14, 1484–1495 (2009).
Nilsson, M. I. et al. Abnormal protein turnover and anabolic resistance to exercise in sarcopenic obesity. FASEB J. 27, 3905–3916 (2013).
Shefer, G., Rauner, G., Stuelsatz, P., Benayahu, D. & Yablonka-Reuveni, Z. Moderate-intensity treadmill running promotes expansion of the satellite cell pool in young and old mice. FEBS J. 280, 4063–4073 (2013).
Zwetsloot, K. A., Childs, T. E., Gilpin, L. T. & Booth, F. W. Non-passaged muscle precursor cells from 32-month old rat skeletal muscle have delayed proliferation and differentiation. Cell Prolif. 46, 45–57 (2013).
Sakuma, K. et al. The adaptive responses in several mediators linked with hypertrophy and atrophy of skeletal muscle after lower limb unloading in humans. Acta Physiol. (Oxf.) 197, 151–159 (2009).
Srikanthan, P., Hevener, A. L. & Karlamangla, A. S. Sarcopenia exacerbates obesity-associated insulin resistance and dysglycemia: findings from the National Health and Nutrition Examination Survey III. PLoS ONE 5, e10805 (2010).
Joseph, A. M., Adhihetty, P. J. & Leeuwenburgh, C. Beneficial effects of exercise on age-related mitochondrial dysfunction and oxidative stress in skeletal muscle. J. Physiol. 594, 5105–5123 (2016).
Schubert, M. M., Sabapathy, S., Leveritt, M. & Desbrow, B. Acute exercise and hormones related to appetite regulation: a meta-analysis. Sports Med. 44, 387–403 (2014).
Roque, F. R. et al. Aerobic exercise reduces oxidative stress and improves vascular changes of small mesenteric and coronary arteries in hypertension. Br. J. Pharmacol. 168, 686–703 (2013).
Kumaraguru, U., Nandakumar, S., Chi, D., Stone, M. & Krishnaswamy, G. Resistance training and aerobic exercise alters immune function (87.25). J. Immunol. 184, 87.25 (2010).
Carraro, F., Stuart, C. A., Hartl, W. H., Rosenblatt, J. & Wolfe, R. R. Effect of exercise and recovery on muscle protein synthesis in human subjects. Am. J. Physiol. 259, E470–E476 (1990).
Drummond, M. J. et al. Rapamycin administration in humans blocks the contraction-induced increase in skeletal muscle protein synthesis. J. Physiol. 587, 1535–1546 (2009).
Fujita, S. et al. Aerobic exercise overcomes the age-related insulin resistance of muscle protein metabolism by improving endothelial function and Akt/mammalian target of rapamycin signaling. Diabetes 56, 1615–1622 (2007).
Levenhagen, D. K. et al. Postexercise nutrient intake timing in humans is critical to recovery of leg glucose and protein homeostasis. Am. J. Physiol. Endocrinol. Metab. 280, E982–E993 (2001).
Sheffield-Moore, M. et al. Postexercise protein metabolism in older and younger men following moderate-intensity aerobic exercise. Am. J. Physiol. Endocrinol. Metab. 287, E513–E522 (2004).
Short, K. R., Vittone, J. L., Bigelow, M. L., Proctor, D. N. & Nair, K. S. Age and aerobic exercise training effects on whole body and muscle protein metabolism. Am. J. Physiol. Endocrinol. Metab. 286, E92–E101 (2004).
Yarasheski, K. E., Zachwieja, J. J. & Bier, D. M. Acute effects of resistance exercise on muscle protein synthesis rate in young and elderly men and women. Am. J. Physiol. 265, E210–E214 (1993).
Yarasheski, K. E. et al. Resistance exercise training increases mixed muscle protein synthesis rate in frail women and men >/=76 yr old. Am. J. Physiol. 277, E118–E125 (1999).
Hasten, D. L., Pak-Loduca, J., Obert, K. A. & Yarasheski, K. E. Resistance exercise acutely increases MHC and mixed muscle protein synthesis rates in 78–84 and 23–32 yr olds. Am. J. Physiol. Endocrinol. Metab. 278, E620–E626 (2000).
Villareal, D. T., Smith, G. I., Sinacore, D. R., Shah, K. & Mittendorfer, B. Regular multicomponent exercise increases physical fitness and muscle protein anabolism in frail, obese, older adults. Obesity (Silver Spring) 19, 312–318 (2011). This study demonstrates that a multicomponent exercise programme consisting of strength, endurance, flexibility and balance increases the basal rate of muscle protein synthesis without affecting the magnitude of the muscle protein anabolic response to feeding.
Cuthbertson, D. et al. Anabolic signaling deficits underlie amino acid resistance of wasting, aging muscle. FASEB J. 19, 422–424 (2005).
Volpi, E., Mittendorfer, B., Rasmussen, B. B. & Wolfe, R. R. The response of muscle protein anabolism to combined hyperaminoacidemia and glucose-induced hyperinsulinemia is impaired in the elderly. J. Clin. Endocrinol. Metab. 85, 4481–4490 (2000). This study highlights the anabolic response to feeding in elderly individuals that underscores sarcopenia of ageing.
Volpi, E., Sheffield-Moore, M., Rasmussen, B. B. & Wolfe, R. R. Basal muscle amino acid kinetics and protein synthesis in healthy young and older men. JAMA 286, 1206–1212 (2001).
Peterson, C. M., Johannsen, D. L. & Ravussin, E. Skeletal muscle mitochondria and aging: a review. J. Aging Res. 2012, 194821 (2012).
Dreyer, H. C., Blanco, C. E., Sattler, F. R., Schroeder, E. T. & Wiswell, R. A. Satellite cell numbers in young and older men 24 hours after eccentric exercise. Muscle Nerve 33, 242–253 (2006).
Petrella, J. K., Kim, J. S., Mayhew, D. L., Cross, J. M. & Bamman, M. M. Potent myofiber hypertrophy during resistance training in humans is associated with satellite cell-mediated myonuclear addition: a cluster analysis. J. Appl. Physiol. (1985) 104, 1736–1742 (2008).
Kang, J. S. & Krauss, R. S. Muscle stem cells in developmental and regenerative myogenesis. Curr. Opin. Clin. Nutr. Metab. Care 13, 243–248 (2010).
Thornell, L. E. Sarcopenic obesity: satellite cells in the aging muscle. Curr. Opin. Clin. Nutr. Metab. Care 14, 22–27 (2011).
Bruunsgaard, H., Bjerregaard, E., Schroll, M. & Pedersen, B. K. Muscle strength after resistance training is inversely correlated with baseline levels of soluble tumor necrosis factor receptors in the oldest old. J. Am. Geriatr. Soc. 52, 237–241 (2004).
Kohut, M. L. et al. Aerobic exercise, but not flexibility/resistance exercise, reduces serum IL-18, CRP, and IL-6 independent of beta-blockers, BMI, and psychosocial factors in older adults. Brain Behav. Immun. 20, 201–209 (2006).
Forti, L. N. et al. Strength training reduces circulating interleukin-6 but not brain-derived neurotrophic factor in community-dwelling elderly individuals. Age (Dordr.) 36, 9704 (2014).
Beyer, I., Mets, T. & Bautmans, I. Chronic low-grade inflammation and age-related sarcopenia. Curr. Opin. Clin. Nutr. Metab. Care 15, 12–22 (2012).
Woods, J. A., Wilund, K. R., Martin, S. A. & Kistler, B. M. Exercise, inflammation and aging. Aging Dis. 3, 130–140 (2012).
Visser, M. et al. Relationship of interleukin-6 and tumor necrosis factor-alpha with muscle mass and muscle strength in elderly men and women: the Health ABC Study. J. Gerontol. A Biol. Sci. Med. Sci. 57, M326–M332 (2002).
Cappola, A. R. et al. Insulin-like growth factor I and interleukin-6 contribute synergistically to disability and mortality in older women. J. Clin. Endocrinol. Metab. 88, 2019–2025 (2003).
Suetta, C. et al. Training-induced changes in muscle CSA, muscle strength, EMG, and rate of force development in elderly subjects after long-term unilateral disuse. J. Appl. Physiol. (1985) 97, 1954–1961 (2004).
Marzetti, E. et al. Modulation of age-induced apoptotic signaling and cellular remodeling by exercise and calorie restriction in skeletal muscle. Free Radic. Biol. Med. 44, 160–168 (2008).
Canto, C. et al. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature 458, 1056–1060 (2009).
Marzetti, E. et al. Physical activity and exercise as countermeasures to physical frailty and sarcopenia. Aging Clin. Exp. Res. 29, 35–42 (2017). This study provides an overview of the interplay between physical activity, exercise and sarcopenia.
Hood, D. A. Mechanisms of exercise-induced mitochondrial biogenesis in skeletal muscle. Appl. Physiol. Nutr. Metab. 34, 465–472 (2009).
Mooren, F. C. & Kruger, K. Exercise, autophagy, and apoptosis. Prog. Mol. Biol. Transl Sci. 135, 407–422 (2015).
Wohlgemuth, S. E. et al. An exploratory analysis of the effects of a weight loss plus exercise program on cellular quality control mechanisms in older overweight women. Rejuvenation Res. 14, 315–324 (2011).
Lambert, C. P. & Evans, W. J. Adaptations to aerobic and resistance exercise in the elderly. Rev. Endocr. Metab. Disord. 6, 137–143 (2005).
Moayedi, Y. et al. The prognostic significance of frailty compared to peak oxygen consumption and B-type natriuretic peptide in patients with advanced heart failure. Clin. Transplant. https://doi.org/10.1111/ctr.13158 (2018).
Jones, S. et al. Assessment of exercise capacity and oxygen consumption using a 6 min stepper test in older adults. Front. Physiol. 8, 408 (2017).
Binder, E. F. et al. Effects of exercise training on frailty in community-dwelling older adults: results of a randomized, controlled trial. J. Am. Geriatr. Soc. 50, 1921–1928 (2002). This is a randomized controlled trial demonstrating that intensive exercise training can improve measures of physical function.
Villareal, D. T., Banks, M., Siener, C., Sinacore, D. R. & Klein, S. Physical frailty and body composition in obese elderly men and women. Obes. Res. 12, 913–920 (2004).
Lambert, C. P., Wright, N. R., Finck, B. N. & Villareal, D. T. Exercise but not diet-induced weight loss decreases skeletal muscle inflammatory gene expression in frail obese elderly persons. J. Appl. Physiol. (1985) 105, 473–478 (2008).
Greiwe, J. S., Cheng, B., Rubin, D. C., Yarasheski, K. E. & Semenkovich, C. F. Resistance exercise decreases skeletal muscle tumor necrosis factor alpha in frail elderly humans. FASEB J. 15, 475–482 (2001).
Gielen, S. et al. Anti-inflammatory effects of exercise training in the skeletal muscle of patients with chronic heart failure. J. Am. Coll. Cardiol. 42, 861–868 (2003).
Markofski, M. M. et al. Exercise training modifies ghrelin and adiponectin concentrations and is related to inflammation in older adults. J. Gerontol. A Biol. Sci. Med. Sci. 69, 675–681 (2014).
Andersen, L. L. et al. Changes in the human muscle force-velocity relationship in response to resistance training and subsequent detraining. J. Appl. Physiol. (1985) 99, 87–94 (2005).
Englund, D. A., Sharp, R. L., Selsby, J. T., Ganesan, S. S. & Franke, W. D. Resistance training performed at distinct angular velocities elicits velocity-specific alterations in muscle strength and mobility status in older adults. Exp. Gerontol. 91, 51–56 (2017).
Fatouros, I. G. et al. Intensity of resistance exercise determines adipokine and resting energy expenditure responses in overweight elderly individuals. Diabetes Care 32, 2161–2167 (2009).
Reeves, N. D., Maganaris, C. N. & Narici, M. V. Effect of strength training on human patella tendon mechanical properties of older individuals. J. Physiol. 548, 971–981 (2003).
Kamen, G. & Knight, C. A. Training-related adaptations in motor unit discharge rate in young and older adults. J. Gerontol. A Biol. Sci. Med. Sci. 59, 1334–1338 (2004).
Prestes, J. et al. Effects of resistance training on resistin, leptin, cytokines, and muscle force in elderly post-menopausal women. J. Sports Sci. 27, 1607–1615 (2009).
Nindl, B. C. et al. Leptin concentrations experience a delayed reduction after resistance exercise in men. Med. Sci. Sports Exerc. 34, 608–613 (2002).
Bouassida, A. et al. Leptin, its implication in physical exercise and training: a short review. J. Sports Sci. Med. 5, 172–181 (2006).
Fatouros, I. G. et al. Leptin and adiponectin responses in overweight inactive elderly following resistance training and detraining are intensity related. J. Clin. Endocrinol. Metab. 90, 5970–5977 (2005).
Rostas, I. et al. In middle-aged and old obese patients, training intervention reduces leptin level: a meta-analysis. PLoS ONE 12, e0182801 (2017).
Nikseresht, M., Sadeghifard, N., Agha-Alinejad, H. & Ebrahim, K. Inflammatory markers and adipocytokine responses to exercise training and detraining in men who are obese. J. Strength Cond. Res. 28, 3399–3410 (2014).
Friedenreich, C. M. et al. Changes in insulin resistance indicators, IGFs, and adipokines in a year-long trial of aerobic exercise in postmenopausal women. Endocr. Relat. Cancer 18, 357–369 (2011).
Haider, S. et al. Change in inflammatory parameters in prefrail and frail persons obtaining physical training and nutritional support provided by lay volunteers: a randomized controlled trial. PLoS ONE 12, e0185879 (2017).
Woodrow, G. Body composition analysis techniques in the aged adult: indications and limitations. Curr. Opin. Clin. Nutr. Metab. Care 12, 8–14 (2009).
Heymsfield, S. B., Gonzalez, M. C., Lu, J., Jia, G. & Zheng, J. Skeletal muscle mass and quality: evolution of modern measurement concepts in the context of sarcopenia. Proc. Nutr. Soc. 74, 355–366 (2015). This is a crucial review that elicits important questions related to loss of lean tissues with dieting.
Baracos, V. et al. Advances in the science and application of body composition measurement. JPEN J. Parenter. Enteral Nutr. 36, 96–107 (2012).
Earthman, C. P. Body composition tools for assessment of adult malnutrition at the bedside: a tutorial on research considerations and clinical applications. JPEN J. Parenter. Enteral Nutr. 39, 787–822 (2015).
Heymsfield, S. B., Gonzalez, M. C., Shen, W., Redman, L. & Thomas, D. Weight loss composition is one-fourth fat-free mass: a critical review and critique of this widely cited rule. Obes. Rev. 15, 310–321 (2014).
Teigen, L. M., Kuchnia, A. J., Mourtzakis, M. & Earthman, C. P. The use of technology for estimating body composition strengths and weaknesses of common modalities in a clinical setting [formula: see text]. Nutr. Clin. Pract. 32, 20–29 (2017).
Cruz-Jentoft, A. J. et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 39, 412–423 (2010).
Kendler, D. L. et al. The Official Positions of the International Society for Clinical Densitometry: indications of use and reporting of DXA for body composition. J. Clin. Densitom. 16, 496–507 (2013).
Fakhrawi, D. H. et al. Comparison of body composition by bioelectrical impedance and dual-energy x-ray absorptiometry in overweight/obese postmenopausal women. J. Clin. Densitom. 12, 238–244 (2009).
Bridge, P. et al. Validation of longitudinal DXA changes in body composition from pre- to mid-adolescence using MRI as reference. J. Clin. Densitom. 14, 340–347 (2011).
Xu, L. et al. Comparisons of body-composition prediction accuracy: a study of 2 bioelectric impedance consumer devices in healthy chinese persons using DXA and MRI as criteria methods. J. Clin. Densitom. 14, 458–464 (2011).
Park, Y. W., Heymsfield, S. B. & Gallagher, D. Are dual-energy X-ray absorptiometry regional estimates associated with visceral adipose tissue mass? Int. J. Obes. Relat. Metab. Disord. 26, 978–983 (2002).
Kaul, S. et al. Dual-energy X-ray absorptiometry for quantification of visceral fat. Obesity 20, 1313–1318 (2012).
Micklesfield, L. K., Goedecke, J. H., Punyanitya, M., Wilson, K. E. & Kelly, T. L. Dual-energy X-ray performs as well as clinical computed tomography for the measurement of visceral fat. Obesity 20, 1109–1114 (2012).
Das, S. K. et al. Body composition assessment in extreme obesity and after massive weight loss induced by gastric bypass surgery. Am. J. Physiol. Endocrinol. Metab. 284, E1080–E1088 (2003).
Schautz, B., Later, W., Heller, M., Muller, M. J. & Bosy-Westphal, A. Total and regional relationship between lean and fat mass with increasing adiposity — impact for the diagnosis of sarcopenic obesity. Eur. J. Clin. Nutr. 66, 1356–1361 (2012).
Bosy-Westphal, A. & Muller, M. J. Identification of skeletal muscle mass depletion across age and BMI groups in health and disease—there is need for a unified definition. Int. J. Obes. (Lond.) 39, 379–386 (2015). This review argues for the importance of standardized definitions in skeletal muscle mass.
Bosy-Westphal, A. et al. Quantification of whole-body and segmental skeletal muscle mass using phase-sensitive 8-electrode medical bioelectrical impedance devices. Eur. J. Clin. Nutr. 71, 1061–1067 (2017).
Houtkooper, L. B., Lohman, T. G., Going, S. B. & Howell, W. H. Why bioelectrical impedance analysis should be used for estimating adiposity. Am. J. Clin. Nutr. 64, 436S–448S (1996).
Kyle, U. G. et al. Bioelectrical impedance analysis-part II: utilization in clinical practice. Clin. Nutr. 23, 1430–1453 (2004). This study demonstrates the application of bioelectrical impedance equations in clinical practice, as advocated by ESPEN.
Chumlea, W. C. et al. Body composition estimates from NHANES III bioelectrical impedance data. Int. J. Obes. Relat. Metab. Disord. 26, 1596–1609 (2002). This study presents normative bioelectrical impedance analysis equations validated using a population-based cohort of American adults.
Janssen, I., Heymsfield, S. B., Baumgartner, R. N. & Ross, R. Estimation of skeletal muscle mass by bioelectrical impedance analysis. J. Appl. Physiol. (1985) 89, 465–471 (2000).
Mally, K., Trentmann, J., Heller, M. & Dittmar, M. Reliability and accuracy of segmental bioelectrical impedance analysis for assessing muscle and fat mass in older Europeans: a comparison with dual-energy X-ray absorptiometry. Eur. J. Appl. Physiol. (1985) 111, 1879–1887 (2011).
Yu, S. C., Powell, A., Khow, K. S. & Visvanathan, R. The performance of five bioelectrical impedance analysis prediction equations against dual x-ray absorptiometry in estimating appendicular skeletal muscle mass in an adult Australian population. Nutrients 8, 189 (2016).
Chien, M. Y., Huang, T. Y. & Wu, Y. T. Prevalence of sarcopenia estimated using a bioelectrical impedance analysis prediction equation in community-dwelling elderly people in Taiwan. J. Am. Geriatr. Soc. 56, 1710–1715 (2008).
Roubenoff, R. Applications of bioelectrical impedance analysis for body composition to epidemiologic studies. Am J. Clin. Nutr. 64, 459S–462S (1996).
Kyle, U. G. et al. Bioelectrical impedance analysis —part I: review of principles and methods. Clin. Nutr. 23, 1226–1243 (2004).
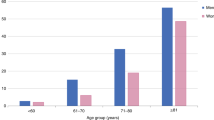
Batsis, J. A., Mackenzie, T. A., Lopez-Jimenez, F. & Bartels, S. J. Sarcopenia, sarcopenic obesity, and functional impairments in older adults: National Health and Nutrition Examination Surveys 1999–2004. Nutr. Res. 35, 1031–1039 (2015).
Batsis, J. A. et al. Variation in the prevalence of sarcopenia and sarcopenic obesity in older adults associated with different research definitions: dual-energy X-ray absorptiometry data from the National Health and Nutrition Examination Survey 1999–2004. J. Am. Geriatr. Soc. 61, 974–980 (2013). This study applies multiple definitions of sarcopenic obesity on a cohort of American adults and emphasizes the varied prevalences of this disorder.
Dehghan, M. & Merchant, A. T. Is bioelectrical impedance accurate for use in large epidemiological studies? Nutr. J. 7, 26 (2008).
Fielding, R. A. et al. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International working group on sarcopenia. J. Am. Med. Dir. Assoc. 12, 249–256 (2011). This is a consensus statement from the International Working Group on Sarcopenia that presents evidence and a definition for the identification of sarcopenia.
Janssen, I., Baumgartner, R. N., Ross, R., Rosenberg, I. H. & Roubenoff, R. Skeletal muscle cutpoints associated with elevated physical disability risk in older men and women. Am. J. Epidemiol. 159, 413–421 (2004).
Studenski, S. A. et al. The FNIH sarcopenia project: rationale, study description, conference recommendations, and final estimates. J. Gerontol. A Biol. Sci. Med. Sci. 69, 547–558 (2014). This report details recommendations from the Foundation of the National Institutes on Health Sarcopenia Project, which aggregated multiple cohorts of older adults and created cut-off points to permit identification of low muscle mass and low muscle strength.
Kemmler, W. et al. Prevalence of sarcopenia and sarcopenic obesity in older German men using recognized definitions: high accordance but low overlap! Osteoporos. Int. 28, 1881–1891 (2017).
Cruz-Jentoft, A. J. et al. Prevalence of and interventions for sarcopenia in ageing adults: a systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing 43, 748–759 (2014).
Chen, L. K. et al. Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J. Am. Med. Dir. Assoc. 15, 95–101 (2014).
Villareal, D. T. et al. Obesity in older adults: technical review and position statement of the American Society for Nutrition and NAASO, The Obesity Society. Am. J. Clin. Nutr. 82, 923–934 (2005). This study highlights current guidelines for the management of older adults with obesity.
Garvey, W. T. et al. American Association of Clinical Endocrinologists and American College of Endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr. Pract. 22 (Suppl. 3), 1–203 (2016). Guidelines for the management of obesity by the AACE/ACE.
Bouchard, D. R., Dionne, I. J. & Brochu, M. Sarcopenic/obesity and physical capacity in older men and women: data from the Nutrition as a Determinant of Successful Aging (NuAge)-the Quebec longitudinal Study. Obesity (Silver Spring) 17, 2082–2088 (2009).
Durstine, J. in ACSM’s Exercise Management for Persons with Chronic Diseases and Disabilities 4th edn (eds Moore, G. E., Durstine, J. L. & Painter, P. L.) (Human Kinetics, 2016).
Baumgartner, R. N. et al. Epidemiology of sarcopenia among the elderly in New Mexico. Am. J. Epidemiol. 147, 755–763 (1998). This is a landmark study that demonstrates the prevalence of sarcopenia in a cohort of older adults in the New Mexico Elder Health Survey.
Baumgartner, R. N. et al. Sarcopenic obesity predicts instrumental activities of daily living disability in the elderly. Obes. Res. 12, 1995–2004 (2004). This study highlights the synergistic problems of sarcopenia and obesity that lead to impairments in instrumental activities of daily living.
Fox, C. S. et al. Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the Framingham Heart Study. Circulation 116, 39–48 (2007).
Despres, J. P. Body fat distribution and risk of cardiovascular disease: an update. Circulation 126, 1301–1313 (2012).
Batsis, J. A. et al. Diagnostic accuracy of body mass index to identify obesity in older adults: NHANES 1999–2004. Int. J. Obes. (Lond.) 40, 761–767 (2016). This study emphasizes the challenges of using BMI as a marker for obesity. BMI has a poor sensitivity in ascertaining body fat-defined obesity that worsens with age.
Schrauwen-Hinderling, V. B., Hesselink, M. K., Schrauwen, P. & Kooi, M. E. Intramyocellular lipid content in human skeletal muscle. Obesity (Silver Spring) 14, 357–367 (2006).
Kemmler, W., von Stengel, S., Engelke, K., Sieber, C. & Freiberger, E. Prevalence of sarcopenic obesity in Germany using established definitions: baseline data of the FORMOsA study. Osteoporos. Int. 27, 275–281 (2016).
Johnson Stoklossa, C. A. et al. Prevalence of sarcopenic obesity in adults with class II/III obesity using different diagnostic criteria. J. Nutr. Metab. 2017, 7307618 (2017).
Batsis, J. A., Mackenzie, T. A., Emeny, R. T., Lopez-Jimenez, F. & Bartels, S. J. Low lean mass with and without obesity, and mortality: results from the 1999–2004 National Health and Nutrition Examination Survey. J. Gerontol. A Biol. Sci. Med. Sci. 72, 1445–1451 (2017). This report details the definition of low lean mass determined by the Foundation of the National Institutes of Health, as well as the adverse association between low lean mass and mortality.
Kim, T. N. et al. Prevalence of sarcopenia and sarcopenic obesity in Korean adults: the Korean sarcopenic obesity study. Int. J. Obes. (Lond.) 33, 885–892 (2009).
Stenholm, S. et al. The effect of obesity combined with low muscle strength on decline in mobility in older persons: results from the InCHIANTI study. Int. J. Obes. (Lond.) 33, 635–644 (2009). This study details data from the InCHIANTI study that prove that obesity combined with low muscle strength increases the risk of mobility disability with age.
Stephen, W. C. & Janssen, I. Sarcopenic-obesity and cardiovascular disease risk in the elderly. J. Nutr. Health Aging 13, 460–466 (2009).
Alley, D. E. et al. Grip strength cutpoints for the identification of clinically relevant weakness. J. Gerontol. A Biol. Sci. Med. Sci. 69, 559–566 (2014).
Rolland, Y. et al. Difficulties with physical function associated with obesity, sarcopenia, and sarcopenic-obesity in community-dwelling elderly women: the EPIDOS (EPIDemiologie de l’OSteoporose) Study. Am. J. Clin. Nutr. 89, 1895–1900 (2009).
Hirani, V. et al. Longitudinal associations between body composition, sarcopenic obesity and outcomes of frailty, disability, institutionalisation and mortality in community-dwelling older men: The Concord Health and Ageing in Men Project. Age Ageing 46, 413–420 (2017).
Zoico, E. et al. Physical disability and muscular strength in relation to obesity and different body composition indexes in a sample of healthy elderly women. Int. J. Obes. Relat. Metab. Disord. 28, 234–241 (2004).
Schaap, L. A., Koster, A. & Visser, M. Adiposity, muscle mass, and muscle strength in relation to functional decline in older persons. Epidemiol. Rev. 35, 51–65 (2013). This is a meta-analysis that proves the longitudinal impact of BMI on incident loss of muscle mass and muscle strength.
Batsis, J. A., Zbehlik, A. J., Pidgeon, D. & Bartels, S. J. Dynapenic obesity and the effect on long-term physical function and quality of life: data from the osteoarthritis initiative. BMC Geriatr. 15, 118 (2015).
Sayers, S. P. et al. Validation of the late-life function and disability instrument. J. Am. Geriatr. Soc. 52, 1554–1559 (2004).
Ware, J. Jr, Kosinski, M. & Keller, S. D. A. 12-item short-form health survey: construction of scales and preliminary tests of reliability and validity. Med. Care 34, 220–233 (1996).
Yang, M., Jiang, J., Hao, Q., Luo, L. & Dong, B. Dynapenic obesity and lower extremity function in elderly adults. J. Am. Med. Dir. Assoc. 16, 31–36 (2015).
Kim, Y. et al. Adiposity and grip strength as long-term predictors of objectively measured physical activity in 93 015 adults: the UK Biobank study. Int. J. Obes. (Lond.) 41, 1361–1368 (2017). This study analyses data from the UK Biobank study and shows that advanced obesity and poor strength at baseline independently predict lower activities at follow-up.
Jung, S. et al. Obesity and muscle weakness as risk factors for mobility limitation in community-dwelling older Japanese women: a two-year follow-up investigation. J. Nutr. Health Aging 20, 28–34 (2016).
Ochi, M. et al. Quadriceps sarcopenia and visceral obesity are risk factors for postural instability in the middle-aged to elderly population. Geriatr. Gerontol. Int. 10, 233–243 (2010).
Baek, S. J. et al. Sarcopenia and sarcopenic obesity and their association with dyslipidemia in Korean elderly men: the 2008–2010 Korea National Health and Nutrition Examination Survey. J. Endocrinol. Invest. 37, 247–260 (2014).
Chung, J. Y., Kang, H. T., Lee, D. C., Lee, H. R. & Lee, Y. J. Body composition and its association with cardiometabolic risk factors in the elderly: a focus on sarcopenic obesity. Arch. Gerontol. Geriatr. 56, 270–278 (2013).
Batsis, J. A., Mackenzie, T. A., Jones, J. D., Lopez-Jimenez, F. & Bartels, S. J. Sarcopenia, sarcopenic obesity and inflammation: results from the 1999–2004 National Health and Nutrition Examination Survey. Clin. Nutr. 35, 1472–1483 (2016).
Aubertin-Leheudre, M., Lord, C., Goulet, E. D., Khalil, A. & Dionne, I. J. Effect of sarcopenia on cardiovascular disease risk factors in obese postmenopausal women. Obesity (Silver Spring) 14, 2277–2283 (2006).
Lee, S., Kim, T. N. & Kim, S. H. Sarcopenic obesity is more closely associated with knee osteoarthritis than is nonsarcopenic obesity: a cross-sectional study. Arthritis Rheum. 64, 3947–3954 (2012).
Scott, D. et al. Sarcopenic obesity and dynapenic obesity: 5-year associations with falls risk in middle-aged and older adults. Obesity (Silver Spring) 22, 1568–1574 (2014).
Scott, D. et al. Associations of sarcopenic obesity and dynapenic obesity with bone mineral density and incident fractures over 5–10 years in community-dwelling older adults. Calcif. Tissue Int. 99, 30–42 (2016).
Atkins, J. L. et al. Sarcopenic obesity and risk of cardiovascular disease and mortality: a population-based cohort study of older men. J. Am. Geriatr. Soc. 62, 253–260 (2014).
Hamer, M., Batty, G. D. & Kivimaki, M. Sarcopenic obesity and risk of new onset depressive symptoms in older adults: English Longitudinal Study of Ageing. Int. J. Obes. (Lond.) 39, 1717–1720 (2015). This study includes longitudinal data that suggest an association between sarcopenic obesity and depression.
Ishii, S. et al. The association between sarcopenic obesity and depressive symptoms in older Japanese adults. PLoS ONE 11, e0162898 (2016).
Mei, K. L., Batsis, J. A., Mills, J. B. & Holubar, S. D. Sarcopenia and sarcopenic obesity: do they predict inferior oncologic outcomes after gastrointestinal cancer surgery? Perioper. Med. (Lond.) 5, 30 (2016).
Androga, L., Sharma, D., Amodu, A. & Abramowitz, M. K. Sarcopenia, obesity, and mortality in US adults with and without chronic kidney disease. Kidney Int. Rep. 2, 201–211 (2017).
Malhotra, R. et al. Sarcopenic obesity definitions by body composition and mortality in the hemodialysis patients. J. Ren. Nutr. 27, 84–90 (2017).
Rossi, A. P. et al. Dynapenic abdominal obesity as a predictor of worsening disability, hospitalization, and mortality in older adults: results from the InCHIANTI Study. J. Gerontol. A Biol. Sci. Med. Sci. 72, 1098–1104 (2017).
Stenholm, S. et al. Obesity and muscle strength as long-term determinants of all-cause mortality — a 33-year follow-up of the Mini-Finland Health Examination Survey. Int. J. Obes. (Lond.) 38, 1126–1132 (2014).
Hamer, M. & O’Donovan, G. Sarcopenic obesity, weight loss, and mortality: the English Longitudinal Study of Ageing. Am. J. Clin. Nutr. 106, 125–129 (2017).
Tian, S. & Xu, Y. Association of sarcopenic obesity with the risk of all-cause mortality: a meta-analysis of prospective cohort studies. Geriatr. Gerontol. Int. 16, 155–166 (2016).
Newman, A. B. et al. Strength, but not muscle mass, is associated with mortality in the health, aging and body composition study cohort. J. Gerontol. A Biol. Sci. Med. Sci. 61, 72–77 (2006). This study demonstrates the importance of muscle strength as opposed to muscle mass as a key determinant of mortality.
Rossi, A. P. et al. Dynapenic abdominal obesity as predictor of mortality and disability worsening in older adults: a 10-year prospective study. Clin. Nutr. 35, 199–204 (2016).
Moon, J. H. et al. Predictive values of the new sarcopenia index by the Foundation for the National Institutes of Health sarcopenia project for mortality among older Korean adults. PLoS ONE 11, e0166344 (2016).
Messier, V. et al. Metabolic profile and quality of life in class I sarcopenic overweight and obese postmenopausal women: a MONET study. Appl. Physiol. Nutr. Metab. 34, 18–24 (2009).
Ware, J. et al. User’s Manual for the SF-36v2 Health Survey (QualityMetric Incorporated, 2007).
Silva Neto, L. S., Karnikowiski, M. G., Tavares, A. B. & Lima, R. M. Association between sarcopenia, sarcopenic obesity, muscle strength and quality of life variables in elderly women. Rev. Bras. Fisioter. 16, 360–367 (2012).
Pedrero-Chamizo, R. et al. Higher levels of physical fitness are associated with a reduced risk of suffering sarcopenic obesity and better perceived health among the elderly: the EXERNET multi-center study. J. Nutr. Health Aging 19, 211–217 (2015).
Cawthon, P. M. et al. Sarcopenia and health care utilization in older women. J. Gerontol. A Biol. Sci. Med. Sci. 72, 95–101 (2017).
Akune, T. et al. Incidence of certified need of care in the long-term care insurance system and its risk factors in the elderly of Japanese population-based cohorts: the ROAD study. Geriatr. Gerontol. Int. 14, 695–701 (2014).
Zizza, C. A., Herring, A., Stevens, J. & Popkin, B. M. Obesity affects nursing-care facility admission among whites but not blacks. Obes. Res. 10, 816–823 (2002).
Elkins, J. S. et al. Midlife obesity and long-term risk of nursing home admission. Obesity (Silver Spring) 14, 1472–1478 (2006).
Valiyeva, E., Russell, L. B., Miller, J. E. & Safford, M. M. Lifestyle-related risk factors and risk of future nursing home admission. Arch. Intern. Med. 166, 985–990 (2006).
Kritchevsky, S. B. et al. Intentional weight loss and all-cause mortality: a meta-analysis of randomized clinical trials. PLoS ONE 10, e0121993 (2015). This is an important meta-analysis that evaluates randomized controlled trials of weight loss interventions in older adults and their impact on mortality.
Batsis, J. A., Huyck, K. L. & Bartels, S. J. Challenges with the Medicare obesity benefit: practical concerns & proposed solutions. J. Gen. Intern. Med. 30, 118–122 (2015).
Jensen, M. D. et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults. Circulation 129, S102–S138 (2014). This report provides clinical obesity guidelines endorsed by multiple national societies.
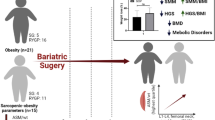
Villareal, D. T. et al. Weight loss, exercise, or both and physical function in obese older adults. N. Engl. J. Med. 364, 1218–1229 (2011). This is a randomized trial of older adults with frailty and obesity that compares diet, exercise type and controls and demonstrates that physical function improves and muscle and bone loss is mitigated with concomitant exercise therapy.
Villareal, D. T. et al. Aerobic or resistance exercise, or both in dieting obese older adults. N. Engl. J. Med. 376, 1943–1955 (2017). This is a randomized trial of older adults with obesity and frailty demonstrating that weight loss coupled with both aerobic and resistance activities is associated with improved physical function.
Chen, H. T., Chung, Y. C., Chen, Y. J., Ho, S. Y. & Wu, H. J. Effects of different types of exercise on body composition, muscle strength, and IGF-1 in the elderly with sarcopenic obesity. J. Am. Geriatr. Soc. 65, 827–832 (2017).
McTigue, K. M. et al. Screening and interventions for obesity in adults: summary of the evidence for the U.S. Preventive Services Task Force. Ann. Intern. Med. 139, 933–949 (2003).
Batsis, J. A. et al. Weight loss interventions in older adults with obesity: a systematic review of randomized controlled trials since 2005. J. Am. Geriatr. Soc. 65, 257–268 (2017). This is a comprehensive systematic review of randomized trials in older adults with obesity highlighting the importance of the improvement in physical function through diet and exercise.
Johnston, B. C. et al. Comparison of weight loss among named diet programs in overweight and obese adults: a meta-analysis. JAMA 312, 923–933 (2014).
Dansinger, M. L., Gleason, J. A., Griffith, J. L., Selker, H. P. & Schaefer, E. J. Comparison of the Atkins, Ornish, Weight Watchers, and Zone diets for weight loss and heart disease risk reduction: a randomized trial. JAMA 293, 43–53 (2005).
Areta, J. L. et al. Reduced resting skeletal muscle protein synthesis is rescued by resistance exercise and protein ingestion following short-term energy deficit. Am. J. Physiol. Endocrinol. Metab. 306, E989–E997 (2014).
Pasiakos, S. M. et al. Effects of high-protein diets on fat-free mass and muscle protein synthesis following weight loss: a randomized controlled trial. FASEB J. 27, 3837–3847 (2013).
Campbell, W. W. et al. Resistance training preserves fat-free mass without impacting changes in protein metabolism after weight loss in older women. Obesity (Silver Spring) 17, 1332–1339 (2009).
Wolfe, R. R., Miller, S. L. & Miller, K. B. Optimal protein intake in the elderly. Clin. Nutr. 27, 675–684 (2008).
Schoenfeld, B. J., Aragon, A. A. & Krieger, J. W. The effect of protein timing on muscle strength and hypertrophy: a meta-analysis. J. Int. Soc. Sports Nutr. 10, 53 (2013).
Smith, G. I. et al. High-protein intake during weight loss therapy eliminates the weight-loss-induced improvement in insulin action in obese postmenopausal women. Cell Rep. 17, 849–861 (2016).
Deutz, N. E. et al. Protein intake and exercise for optimal muscle function with aging: recommendations from the ESPEN Expert Group. Clin. Nutr. 33, 929–936 (2014). These are the European Society for Clinical Nutrition and Metabolism workshop recommendations on protein requirements in elderly individuals.
Bouillanne, O. et al. Impact of protein pulse feeding on lean mass in malnourished and at-risk hospitalized elderly patients: a randomized controlled trial. Clin. Nutr. 32, 186–192 (2013).
Beasley, J. M. et al. Protein intake and incident frailty in the Women’s Health Initiative observational study. J. Am. Geriatr. Soc. 58, 1063–1071 (2010).
Tieland, M. et al. Protein supplementation improves physical performance in frail elderly people: a randomized, double-blind, placebo-controlled trial. J. Am. Med. Dir. Assoc. 13, 720–726 (2012).
Porter Starr, K. N. et al. Improved function with enhanced protein intake per meal: a pilot study of weight reduction in frail, obese older adults. J. Gerontol. A Biol. Sci. Med. Sci. 71, 1369–1375 (2016).
Verreijen, A. M. et al. Effect of a high protein diet and/or resistance exercise on the preservation of fat free mass during weight loss in overweight and obese older adults: a randomized controlled trial. Nutr. J. 16, 10 (2017).
Sammarco, R. et al. Evaluation of hypocaloric diet with protein supplementation in middle-aged sarcopenic obese women: a pilot study. Obes. Facts 10, 160–167 (2017).
Avenell, A. & Handoll, H. H. Nutritional supplementation for hip fracture aftercare in older people. Cochrane Database Syst. Rev. 1, CD001880 (2010).
Cawood, A. L., Elia, M. & Stratton, R. J. Systematic review and meta-analysis of the effects of high protein oral nutritional supplements. Ageing Res. Rev. 11, 278–296 (2012).
Milne, A. C., Potter, J., Vivanti, A. & Avenell, A. Protein and energy supplementation in elderly people at risk from malnutrition. Cochrane Database Syst. Rev. 2, CD003288 (2009). This is a systematic Cochrane review of randomized and quasi-randomized controlled trials of oral protein and energy supplementation in older people, which shows a small but consistent weight gain in older people.
Brenner, B. M., Meyer, T. W. & Hostetter, T. H. Dietary protein intake and the progressive nature of kidney disease: the role of hemodynamically mediated glomerular injury in the pathogenesis of progressive glomerular sclerosis in aging, renal ablation, and intrinsic renal disease. N. Engl. J. Med. 307, 652–659 (1982).
Klahr, S. The modification of diet in renal disease study. N. Engl. J. Med. 320, 864–866 (1989).
Lentine, K. & Wrone, E. M. New insights into protein intake and progression of renal disease. Curr. Opin. Nephrol. Hypertens. 13, 333–336 (2004).
Pedrini, M. T., Levey, A. S., Lau, J., Chalmers, T. C. & Wang, P. H. The effect of dietary protein restriction on the progression of diabetic and nondiabetic renal diseases: a meta-analysis. Ann. Intern. Med. 124, 627–632 (1996).
Bhasin, S. et al. Effect of protein intake on lean body mass in functionally limited older men: a randomized clinical trial. JAMA Intern. Med. 178, 530–541 (2018).
Garber, C. E. et al. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med. Sci. Sports Exerc. 43, 1334–1359 (2011).
Gillespie, L. D. et al. Interventions for preventing falls in older people living in the community. Cochrane Database Syst. Rev. 9, CD007146 (2012).
Lin, X. et al. Effects of exercise training on cardiorespiratory fitness and biomarkers of cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials. J. Am. Heart Assoc. 4, e002014 (2015).
Hwang, C. L., Wu, Y. T. & Chou, C. H. Effect of aerobic interval training on exercise capacity and metabolic risk factors in people with cardiometabolic disorders: a meta-analysis. J. Cardiopulm. Rehabil. Prev. 31, 378–385 (2011).
Sui, X. M. et al. Cardiorespiratory fitness and adiposity as mortality predictors in older adults. JAMA 298, 2507–2516 (2007).
Taaffe, D. R., Duret, C., Wheeler, S. & Marcus, R. Once-weekly resistance exercise improves muscle strength and neuromuscular performance in older adults. J. Am. Geriatr. Soc. 47, 1208–1214 (1999).
Brown, A. B., McCartney, N. & Sale, D. G. Positive adaptations to weight-lifting training in the elderly. J. Appl. Physiol. (1985) 69, 1725–1733 (1990).
Liu, C. J. & Latham, N. K. Progressive resistance strength training for improving physical function in older adults. Cochrane Database Syst. Rev. 3, CD002759 (2009). This is a systematic Cochrane review of randomized controlled trials reporting physical outcomes of progressive resistance therapy that demonstrates improved strength and performance.
Pahor, M. et al. Effect of structured physical activity on prevention of major mobility disability in older adults: the LIFE study randomized clinical trial. JAMA 311, 2387–2396 (2014). This study reports on a gold standard, structured, long-term physical activity intervention, consisting of 3–4 times of aerobic, resistance and flexibility training per week, that demonstrates improvements in mobility disability over a 2.6-year follow-up.
Stec, M. J. et al. Randomized, four-arm, dose-response clinical trial to optimize resistance exercise training for older adults with age-related muscle atrophy. Exp. Gerontol. 99, 98–109 (2017).
Guralnik, J. M., Ferrucci, L., Simonsick, E. M., Salive, M. E. & Wallace, R. B. Lower-extremity function in persons over the age of 70 years as a predictor of subsequent disability. N. Engl. J. Med. 332, 556–561 (1995).
Liu, C. K. et al. The impact of sarcopenia on a physical activity intervention: the Lifestyle Interventions and Independence for Elders Pilot Study (LIFE-P). J. Nutr. Health Aging 18, 59–64 (2014).
Villanueva, M. G., Lane, C. J. & Schroeder, E. T. Short rest interval lengths between sets optimally enhance body composition and performance with 8 weeks of strength resistance training in older men. Eur. J. Appl. Physiol. 115, 295–308 (2015).
Ramirez-Campillo, R. et al. High-speed resistance training is more effective than low-speed resistance training to increase functional capacity and muscle performance in older women. Exp. Gerontol. 58, 51–57 (2014).
Ramirez-Campillo, R. et al. Effects of different doses of high-speed resistance training on physical performance and quality of life in older women: a randomized controlled trial. Clin. Interv. Aging 11, 1797–1804 (2016).
Balachandran, A., Krawczyk, S. N., Potiaumpai, M. & Signorile, J. F. High-speed circuit training versus hypertrophy training to improve physical function in sarcopenic obese adults: a randomized controlled trial. Exp. Gerontol. 60, 64–71 (2014).
Vasconcelos, K. S. et al. Effects of a progressive resistance exercise program with high-speed component on the physical function of older women with sarcopenic obesity: a randomized controlled trial. Braz. J. Phys. Ther. 20, 432–440 (2016).
Son, N. K., Ryu, Y. U., Jeong, H. W., Jang, Y. H. & Kim, H. D. Comparison of 2 different exercise approaches: Tai Chi versus Otago, in community-dwelling older women. J. Geriatr. Phys. Ther. 39, 51–57 (2016).
Huang, Z. G., Feng, Y. H., Li, Y. H. & Lv, C. S. Systematic review and meta-analysis: Tai Chi for preventing falls in older adults. BMJ Open 7, e013661 (2017).
Youkhana, S., Dean, C. M., Wolff, M., Sherrington, C. & Tiedemann, A. Yoga-based exercise improves balance and mobility in people aged 60 and over: a systematic review and meta-analysis. Age Ageing 45, 21–29 (2016).
Waller, B. et al. The effect of aquatic exercise on physical functioning in the older adult: a systematic review with meta-analysis. Age Ageing 45, 593–601 (2016).
Nobrega, S. R. & Libardi, C. A. Is resistance training to muscular failure necessary? Front. Physiol. 7, 10 (2016).
Aguirre, L. E. & Villareal, D. T. Physical exercise as therapy for frailty. Nestle Nutr. Inst. Workshop Ser. 83, 83–92 (2015).
Liao, C. D. et al. Effects of elastic resistance exercise on body composition and physical capacity in older women with sarcopenic obesity: a CONSORT-compliant prospective randomized controlled trial. Medicine (Baltimore) 96, e7115 (2017).
Weinheimer, E. M., Sands, L. P. & Campbell, W. W. A systematic review of the separate and combined effects of energy restriction and exercise on fat-free mass in middle-aged and older adults: implications for sarcopenic obesity. Nutr. Rev. 68, 375–388 (2010). This is a systematic review assessing the effects of energy restriction and exercise on fat-free mass and suggests that exercise is an effective tool in combating sarcopenia-induced weight loss.
Armamento-Villareal, R., Aguirre, L. E., Qualls, C. & Villareal, D. T. Effect of lifestyle intervention on the hormonal profile of frail, obese older men. J. Nutr. Health Aging 20, 334–340 (2016).
Villareal, D. T. et al. Effect of lifestyle intervention on metabolic coronary heart disease risk factors in obese older adults. Am. J. Clin. Nutr. 84, 1317–1323 (2006).
Bouchonville, M. et al. Weight loss, exercise or both and cardiometabolic risk factors in obese older adults: results of a randomized controlled trial. Int. J. Obes. (Lond.) 38, 423–431 (2014).
Abbenhardt, C. et al. Effects of individual and combined dietary weight loss and exercise interventions in postmenopausal women on adiponectin and leptin levels. J. Intern. Med. 274, 163–175 (2013).
Beavers, K. M. et al. Long-term physical activity and inflammatory biomarkers in older adults. Med. Sci. Sports Exerc. 42, 2189–2196 (2010).
Kelly, K. R. et al. Lifestyle-induced decrease in fat mass improves adiponectin secretion in obese adults. Med. Sci. Sports Exerc. 46, 920–926 (2014).
Wang, X., You, T., Murphy, K., Lyles, M. F. & Nicklas, B. J. Addition of exercise increases plasma adiponectin and release from adipose tissue. Med. Sci. Sports Exerc. 47, 2450–2455 (2015).
O’Leary, V. B. et al. Enhanced adiponectin multimer ratio and skeletal muscle adiponectin receptor expression following exercise training and diet in older insulin-resistant adults. Am. J. Physiol. Endocrinol. Metab. 293, E421–E427 (2007).
Nicklas, B. J. et al. Diet-induced weight loss, exercise, and chronic inflammation in older, obese adults: a randomized controlled clinical trial. Am. J. Clin. Nutr. 79, 544–551 (2004). This is an early trial suggesting that resistance training is important for improving body composition and muscle strength in elderly individuals who are obese, an effect which is improved with caloric restriction.
Beavers, K. M., Ambrosius, W. T., Nicklas, B. J. & Rejeski, W. J. Independent and combined effects of physical activity and weight loss on inflammatory biomarkers in overweight and obese older adults. J. Am. Geriatr. Soc. 61, 1089–1094 (2013).
Zibellini, J. et al. Effect of diet-induced weight loss on muscle strength in adults with overweight or obesity — a systematic review and meta-analysis of clinical trials. Obes. Rev. 17, 647–663 (2016).
Gallagher, D. et al. Changes in skeletal muscle and organ size after a weight-loss intervention in overweight and obese type 2 diabetic patients. Am. J. Clin. Nutr. 105, 78–84 (2017).
Weiss, E. P., Jordan, R. C., Frese, E. M., Albert, S. G. & Villareal, D. T. Effects of weight loss on lean mass, strength, bone, and aerobic capacity. Med. Sci. Sports Exerc. 49, 206–217 (2017).
Wood, R. J. et al. Preservation of fat-free mass after two distinct weight loss diets with and without progressive resistance exercise. Metab. Syndr. Relat. Disord. 10, 167–174 (2012).
Tang, X. et al. Obesity and risk of hip fracture in adults: a meta-analysis of prospective cohort studies. PLoS ONE 8, e55077 (2013).
Nielson, C. M. et al. BMI and fracture risk in older men: the osteoporotic fractures in men study (MrOS). J. Bone Miner. Res. 26, 496–502 (2011).
Compston, J. E. et al. Obesity is not protective against fracture in postmenopausal women: GLOW. Am. J. Med. 124, 1043–1050 (2011).
Ensrud, K. E. et al. A comparison of frailty indexes for the prediction of falls, disability, fractures, and mortality in older men. J. Am. Geriatr. Soc. 57, 492–498 (2009).
Sundh, D. et al. A high amount of local adipose tissue is associated with high cortical porosity and low bone material strength in older women. J. Bone Miner. Res. 31, 749–757 (2016).
Zibellini, J. et al. Does diet-induced weight loss lead to bone loss in overweight or obese adults? A systematic review and meta-analysis of clinical trials. J. Bone Miner. Res. 30, 2168–2178 (2015).
Soltani, S., Hunter, G. R., Kazemi, A. & Shab-Bidar, S. The effects of weight loss approaches on bone mineral density in adults: a systematic review and meta-analysis of randomized controlled trials. Osteoporos. Int. 27, 2655–2671 (2016).
Colleluori, G., Napoli, N., Phadnis, U., Armamento-Villareal, R. & Villareal, D. T. Effect of weight loss, exercise, or both on undercarboxylated osteocalcin and insulin secretion in frail, obese older adults. Oxid. Med. Cell. Longev. 2017, 4807046 (2017).
Waters, D. L. et al. Long-term maintenance of weight loss after lifestyle intervention in frail, obese older adults. J. Nutr. Health Aging 17, 3–7 (2013).
Villareal, D. T. et al. Effect of two-year caloric restriction on bone metabolism and bone mineral density in non-obese younger adults: a randomized clinical trial. J. Bone Miner. Res. 31, 40–51 (2016). This is a 2-year randomized trial of caloric restriction, which had been previously shown to lead to bone loss at important sites of osteoporotic fractures as represented by reductions in BMD.
Villareal, D. T. et al. Reduced bone mineral density is not associated with significantly reduced bone quality in men and women practicing long-term calorie restriction with adequate nutrition. Aging Cell 10, 96–102 (2011).
Pop, L. C. et al. Moderate weight loss in obese and overweight men preserves bone quality. Am. J. Clin. Nutr. 101, 659–667 (2015).
Johnson, K. C. et al. The effect of intentional weight loss on fracture risk in persons with diabetes: results from the Look AHEAD randomized clinical trial. J. Bone Miner. Res. 32, 2278–2287 (2017).
Haywood, C. J. et al. Very low calorie diets for weight loss in obese older adults—a randomized trial. J. Gerontol. A Biol. Sci. Med. Sci. 73, 59–65 (2017).
Shah, K. et al. Exercise training in obese older adults prevents increase in bone turnover and attenuates decrease in hip bone mineral density induced by weight loss despite decline in bone-active hormones. J. Bone Miner. Res. 26, 2851–2859 (2011).
Armamento-Villareal, R. et al. Weight loss in obese older adults increases serum sclerostin and impairs hip geometry but both are prevented by exercise training. J. Bone Miner. Res. 27, 1215–1221 (2012).
Moyer, V. A. Vitamin D and calcium supplementation to prevent fractures in adults: U.S. Preventive Services Task Force recommendation statement. Ann. Intern. Med. 158, 691–696 (2013).
Anderson, J. J. et al. Calcium intake from diet and supplements and the risk of coronary artery calcification and its progression among older adults: 10-year follow-up of the multi-ethnic study of atherosclerosis (MESA). J. Am. Heart Assoc. 5, e003815 (2016).
Hassan-Smith, Z. K. et al. 25-hydroxyvitamin D3 and 1,25-dihydroxyvitamin D3 exert distinct effects on human skeletal muscle function and gene expression. PLoS ONE 12, e0170665 (2017).
Beaudart, C. et al. The effects of vitamin D on skeletal muscle strength, muscle mass, and muscle power: a systematic review and meta-analysis of randomized controlled trials. J. Clin. Endocrinol. Metab. 99, 4336–4345 (2014).
Malafarina, V., Uriz-Otano, F., Malafarina, C., Martinez, J. A. & Zulet, M. A. Effectiveness of nutritional supplementation on sarcopenia and recovery in hip fracture patients. A multi-centre randomized trial. Maturitas 101, 42–50 (2017).
American Geriatrics Society Workgroup on Vitamin D Supplementation for Older Adults. Recommendations abstracted from the American Geriatrics Society Consensus Statement on vitamin D for prevention of falls and their consequences. J. Am. Geriatr. Soc. 62, 147–152 (2014).
Bischoff-Ferrari, H. A. et al. Higher 25-hydroxyvitamin D concentrations are associated with better lower-extremity function in both active and inactive persons aged > or =60 y. Am. J. Clin. Nutr. 80, 752–758 (2004).
Scott, D. et al. A prospective study of the associations between 25-hydroxy-vitamin D, sarcopenia progression and physical activity in older adults. Clin. Endocrinol. (Oxf.) 73, 581–587 (2010).
Wicherts, I. S. et al. Vitamin D status predicts physical performance and its decline in older persons. J. Clin. Endocrinol. Metab. 92, 2058–2065 (2007).
Zittermann, A. et al. Vitamin D deficiency and mortality risk in the general population: a meta-analysis of prospective cohort studies. Am. J. Clin. Nutr. 95, 91–100 (2012).
Anker, S. D., Morley, J. E. & von Haehling, S. Welcome to the ICD-10 code for sarcopenia. J. Cachexia Sarcopenia Muscle 7, 512–514 (2016).
Cao, B. Future healthy life expectancy among older adults in the US: a forecast based on cohort smoking and obesity history. Popul. Health Metr. 14, 23 (2016).
U.S. Preventive Services Task Force. Screening for osteoporosis: U.S. preventive services task force recommendation statement. Ann. Intern. Med. 154, 356–364 (2011).
Maddalo, M. et al. Validation of a free software for unsupervised assessment of abdominal fat in MRI. Phys. Med. 37, 24–31 (2017).
van Vugt, J. L. A. et al. A comparative study of software programmes for cross-sectional skeletal muscle and adipose tissue measurements on abdominal computed tomography scans of rectal cancer patients. J. Cachexia Sarcopenia Muscle 8, 285–297 (2017).
Curtis, J. R. et al. The geographic availability and associated utilization of dual-energy X-ray absorptiometry (DXA) testing among older persons in the United States. Osteoporos. Int. 20, 1553–1561 (2009).
Maggio, M. et al. Instrumental and non-instrumental evaluation of 4-meter walking speed in older individuals. PLoS ONE 11, e0153583 (2016).
Nascimento, L. R. et al. Different instructions during the ten-meter walking test determined significant increases in maximum gait speed in individuals with chronic hemiparesis. Rev. Bras. Fisioter. 16, 122–127 (2012).
Roberts, H. C. et al. A review of the measurement of grip strength in clinical and epidemiological studies: towards a standardised approach. Age Ageing 40, 423–429 (2011).
Witard, O. C., McGlory, C., Hamilton, D. L. & Phillips, S. M. Growing older with health and vitality: a nexus of physical activity, exercise and nutrition. Biogerontology 17, 529–546 (2016).
Garatachea, N. et al. Exercise attenuates the major hallmarks of aging. Rejuvenation Res. 18, 57–89 (2015).
Emmelot-Vonk, M. H. et al. Effect of testosterone supplementation on functional mobility, cognition, and other parameters in older men: a randomized controlled trial. JAMA 299, 39–52 (2008).
Travison, T. G. et al. Clinical meaningfulness of the changes in muscle performance and physical function associated with testosterone administration in older men with mobility limitation. J. Gerontol. A Biol. Sci. Med. Sci. 66, 1090–1099 (2011).
Nguyen, H. Q. et al. Health care use and costs associated with use of a health club membership benefit in older adults with diabetes. Diabetes Care 31, 1562–1567 (2008).
Bergamin, M. et al. Water- versus land-based exercise in elderly subjects: effects on physical performance and body composition. Clin. Interv. Aging 8, 1109–1117 (2013).
de Souza Vasconcelos, K. S. et al. Land-based versus aquatic resistance therapeutic exercises for older women with sarcopenic obesity: study protocol for a randomised controlled trial. Trials 14, 296 (2013).
Simmons, V. & Hansen, P. D. Effectiveness of water exercise on postural mobility in the well elderly: an experimental study on balance enhancement. J. Gerontol. A Biol. Sci. Med. Sci. 51, M233–M238 (1996).
Li, F. et al. An evaluation of the effects of Tai Chi exercise on physical function among older persons: a randomized contolled trial. Ann. Behav. Med. 23, 139–146 (2001).
Hildreth, K. L. et al. Effects of testosterone and progressive resistance exercise in healthy, highly functioning older men with low-normal testosterone levels. J. Clin. Endocrinol. Metab. 98, 1891–1900 (2013).
Strohacker, K., Fazzino, D., Breslin, W. L. & Xu, X. The use of periodization in exercise prescriptions for inactive adults: a systematic review. Prev. Med. Rep. 2, 385–396 (2015).
Steele, J. Intensity; in-ten-si-ty; noun. 1. Often used ambiguously within resistance training. 2. Is it time to drop the term altogether? Br. J. Sports Med. 48, 1586–1588 (2014).
Conlon, J. A. et al. Periodization strategies in older adults: impact on physical function and health. Med. Sci. Sports Exerc. 48, 2426–2436 (2016).
Prestes, J. et al. Understanding the individual responsiveness to resistance training periodization. Age (Dordr.) 37, 9793 (2015).
Botero, J. P. et al. Effects of long-term periodized resistance training on body composition, leptin, resistin and muscle strength in elderly post-menopausal women. J. Sports Med. Phys. Fitness 53, 289–294 (2013).
Ebersbach, G., Edler, D., Kaufhold, O. & Wissel, J. Whole body vibration versus conventional physiotherapy to improve balance and gait in Parkinson’s disease. Arch. Phys. Med. Rehabil. 89, 399–403 (2008).
Zhang, L. et al. Effect of whole-body vibration exercise on mobility, balance ability and general health status in frail elderly patients: a pilot randomized controlled trial. Clin. Rehabil. 28, 59–68 (2014).
Rogan, S., Hilfiker, R., Herren, K., Radlinger, L. & de Bruin, E. D. Effects of whole-body vibration on postural control in elderly: a systematic review and meta-analysis. BMC Geriatr. 11, 72 (2011).
Rogan, S. et al. Feasibility and effects of applying stochastic resonance whole-body vibration on untrained elderly: a randomized crossover pilot study. BMC Geriatr. 15, 25 (2015).
Verschueren, S. M. et al. The effects of whole-body vibration training and vitamin D supplementation on muscle strength, muscle mass, and bone density in institutionalized elderly women: a 6-month randomized, controlled trial. J. Bone Miner. Res. 26, 42–49 (2011).
Cardim, A. B. et al. Does whole-body vibration improve the functional exercise capacity of subjects with COPD? A meta-analysis. Respir. Care 61, 1552–1559 (2016).
Burke, D. & Schiller, H. H. Discharge pattern of single motor units in the tonic vibration reflex of human triceps surae. J. Neurol. Neurosurg. Psychiatry 39, 729–741 (1976).
Chen, H., Ma, J., Lu, B. & Ma, X. L. The effect of whole-body vibration training on lean mass: a PRISMA-compliant meta-analysis. Medicine (Baltimore) 96, e8390 (2017).
Lau, R. W. et al. The effects of whole body vibration therapy on bone mineral density and leg muscle strength in older adults: a systematic review and meta-analysis. Clin. Rehabil. 25, 975–988 (2011).
Bogaerts, A. et al. Impact of whole-body vibration training versus fitness training on muscle strength and muscle mass in older men: a 1-year randomized controlled trial. J. Gerontol. A Biol. Sci. Med. Sci. 62, 630–635 (2007).
Chang, S. F., Lin, P. C., Yang, R. S. & Yang, R. J. The preliminary effect of whole-body vibration intervention on improving the skeletal muscle mass index, physical fitness, and quality of life among older people with sarcopenia. BMC Geriatr. 18, 17 (2018).
Bemben, D. A., Palmer, I. J., Bemben, M. G. & Knehans, A. W. Effects of combined whole-body vibration and resistance training on muscular strength and bone metabolism in postmenopausal women. Bone 47, 650–656 (2010).
Fjeldstad, C., Palmer, I. J., Bemben, M. G. & Bemben, D. A. Whole-body vibration augments resistance training effects on body composition in postmenopausal women. Maturitas 63, 79–83 (2009).
Machado, A., Garcia-Lopez, D., Gonzalez-Gallego, J. & Garatachea, N. Whole-body vibration training increases muscle strength and mass in older women: a randomized-controlled trial. Scand. J. Med. Sci. Sports 20, 200–207 (2010).
von Stengel, S., Kemmler, W., Engelke, K. & Kalender, W. A. Effect of whole-body vibration on neuromuscular performance and body composition for females 65 years and older: a randomized-controlled trial. Scand. J. Med. Sci. Sports 22, 119–127 (2012).
Cristi-Montero, C., Cuevas, M. J. & Collado, P. S. Whole-body vibration training as complement to programs aimed at weight loss. Nutr. Hosp. 28, 1365–1371 (2013).
Lamont, H. S. et al. Effects of a 6-week periodized squat training with or without whole-body vibration upon short-term adaptations in squat strength and body composition. J. Strength Cond. Res. 25, 1839–1848 (2011).
Wei, N., Pang, M. Y., Ng, S. S. & Ng, G. Y. Optimal frequency/time combination of whole-body vibration training for improving muscle size and strength of people with age-related muscle loss (sarcopenia): a randomized controlled trial. Geriatr. Gerontol. Int. 17, 1412–1420 (2017).
Perna, S. et al. Liraglutide and obesity in elderly: efficacy in fat loss and safety in order to prevent sarcopenia. A perspective case series study. Aging Clin. Exp. Res. 28, 1251–1257 (2016).
Apovian, C., Palmer, K., Fain, R., Perdomo, C. & Rubino, D. Effects of lorcaserin on fat and lean mass loss in obese and overweight patients without and with type 2 diabetes mellitus: the BLOSSOM and BLOOM-DM studies. Diabetes Obes. Metab. 18, 945–948 (2016).
Simko, J. et al. The effect of topiramate and lamotrigine on rat bone mass, structure and metabolism. J. Neurol. Sci. 340, 80–85 (2014).
Eliasson, B. et al. Weight loss and metabolic effects of topiramate in overweight and obese type 2 diabetic patients: randomized double-blind placebo-controlled trial. Int. J. Obes. (Lond.) 31, 1140–1147 (2007).
Yaman, M. et al. Effects of topiramate use on body composition and resting metabolic rate in migraine patients. Neurol. Sci. 34, 225–229 (2013).
Cordero-MacIntyre, Z. R. et al. Effect of a weight-reduction program on total and regional body composition in obese postmenopausal women. Ann. NY Acad. Sci. 904, 526–535 (2000).
Gadde, K. M., Zhang, W. & Foust, M. S. Bupropion treatment of olanzapine-associated weight gain: an open-label, prospective trial. J. Clin. Psychopharmacol. 26, 409–413 (2006).
Smith, S. R. et al. Combination therapy with naltrexone and bupropion for obesity reduces total and visceral adiposity. Diabetes Obes. Metab. 15, 863–866 (2013).
Kujawska-Luczak, M. et al. The effect of orlistat versus metformin on body composition and insulin resistance in obese premenopausal women: 3-month randomized prospective open-label study. Arch. Med. Sci. 13, 725–731 (2017).
Smith, S. R. et al. Orlistat 60 mg reduces visceral adipose tissue: a 24-week randomized, placebo-controlled, multicenter trial. Obesity (Silver Spring) 19, 1796–1803 (2011).
Smith, T. J. et al. Efficacy of orlistat 60 mg on weight loss and body fat mass in US Army soldiers. J. Acad. Nutr. Diet. 112, 533–540 (2012).
Batsis, J. A. & Dolkart, K. M. Evaluation of older adults with obesity for bariatric surgery: geriatricians’ perspective. J. Clin. Gerontol. Geriatr. 6, 45–53 (2015).
Mastino, D. et al. Bariatric surgery outcomes in sarcopenic obesity. Obes. Surg. 26, 2355–2362 (2016).
Cole, A. J. et al. Long-term body composition changes in women following roux-en-Y gastric bypass surgery. JPEN J. Parenter. Enteral Nutr. 41, 583–591 (2017).
Anderwald, C. H. et al. Alterations in gastrointestinal, endocrine, and metabolic processes after bariatric Roux-en-Y gastric bypass surgery. Diabetes Care 35, 2580–2587 (2012).
Lyytinen, T., Liikavainio, T., Paakkonen, M., Gylling, H. & Arokoski, J. P. Physical function and properties of quadriceps femoris muscle after bariatric surgery and subsequent weight loss. J. Musculoskelet. Neuronal Interact. 13, 329–338 (2013).
Thibault, R., Huber, O., Azagury, D. E. & Pichard, C. Twelve key nutritional issues in bariatric surgery. Clin. Nutr. 35, 12–17 (2016).
Vaurs, C. et al. Determinants of changes in muscle mass after bariatric surgery. Diabetes Metab. 41, 416–421 (2015).
Coates, P. S., Fernstrom, J. D., Fernstrom, M. H., Schauer, P. R. & Greenspan, S. L. Gastric bypass surgery for morbid obesity leads to an increase in bone turnover and a decrease in bone mass. J. Clin. Endocrinol. Metab. 89, 1061–1065 (2004).
Ko, B. J. et al. Relationship between bariatric surgery and bone mineral density: a meta-analysis. Obes. Surg. 26, 1414–1421 (2016).
Shanbhogue, V. V. et al. Bone structural changes after gastric bypass surgery evaluated by HR-pQCT: a two-year longitudinal study. Eur. J. Endocrinol. 176, 685–693 (2017).
Yu, E. W., Lee, M. P., Landon, J. E., Lindeman, K. G. & Kim, S. C. Fracture risk after bariatric surgery: roux-en-Y gastric bypass versus adjustable gastric banding. J. Bone Miner. Res. 32, 1229–1236 (2017).
Cohen, P. G. Obesity in men: the hypogonadal-estrogen receptor relationship and its effect on glucose homeostasis. Med. Hypotheses 70, 358–360 (2008).
Freeman, E. W., Sammel, M. D., Lin, H. & Gracia, C. R. Obesity and reproductive hormone levels in the transition to menopause. Menopause 17, 718–726 (2010).
Nettleship, J. E., Pugh, P. J., Channer, K. S., Jones, T. & Jones, R. D. Inverse relationship between serum levels of interleukin-1beta and testosterone in men with stable coronary artery disease. Horm. Metab. Res. 39, 366–371 (2007).
Corrales, J. J. et al. Androgen-replacement therapy depresses the ex vivo production of inflammatory cytokines by circulating antigen-presenting cells in aging type-2 diabetic men with partial androgen deficiency. J. Endocrinol. 189, 595–604 (2006).
Neto, W. K. et al. Effects of testosterone on lean mass gain in elderly men: systematic review with meta-analysis of controlled and randomized studies. Age (Dordr.) 37, 9742 (2015). This is a meta-analysis demonstrating that testosterone supplementation in elderly men leads to increases in lean mass and potentially reductions in fat mass.
Giannoulis, M. G. et al. The effects of growth hormone and/or testosterone in healthy elderly men: a randomized controlled trial. J. Clin. Endocrinol. Metab. 91, 477–484 (2006).
Schroeder, E. T. et al. Value of measuring muscle performance to assess changes in lean mass with testosterone and growth hormone supplementation. Eur. J. Appl. Physiol. 112, 1123–1131 (2012).
Hyde, Z. et al. Low free testosterone predicts frailty in older men: the health in men study. J. Clin. Endocrinol. Metab. 95, 3165–3172 (2010).
Krasnoff, J. B. et al. Free testosterone levels are associated with mobility limitation and physical performance in community-dwelling men: the Framingham Offspring Study. J. Clin. Endocrinol. Metab. 95, 2790–2799 (2010).
Storer, T. W. et al. Effects of testosterone supplementation for 3 years on muscle performance and physical function in older men. J. Clin. Endocrinol. Metab. 102, 583–593 (2017).
Saad, F., Rohrig, G., von Haehling, S. & Traish, A. Testosterone deficiency and testosterone treatment in older men. Gerontology 63, 144–156 (2017).
Bhasin, S. et al. Older men are as responsive as young men to the anabolic effects of graded doses of testosterone on the skeletal muscle. J. Clin. Endocrinol. Metab. 90, 678–688 (2005). This is a randomized trial of dose-dependent testosterone supplementation that demonstrates that older men are as responsive as young men to the anabolic effects of testosterone; however, older men have lower testosterone clearance rates, higher increments in haemoglobin and a higher frequency of adverse effects.
Srinivas-Shankar, U. et al. Effects of testosterone on muscle strength, physical function, body composition, and quality of life in intermediate-frail and frail elderly men: a randomized, double-blind, placebo-controlled study. J. Clin. Endocrinol. Metab. 95, 639–650 (2010).
Page, S. T. et al. Exogenous testosterone (T) alone or with finasteride increases physical performance, grip strength, and lean body mass in older men with low serum T. J. Clin. Endocrinol. Metab. 90, 1502–1510 (2005).
Ottenbacher, K. J., Ottenbacher, M. E., Ottenbacher, A. J., Acha, A. A. & Ostir, G. V. Androgen treatment and muscle strength in elderly men: a meta-analysis. J. Am. Geriatr. Soc. 54, 1666–1673 (2006).
O’Connell, M. D. et al. Do the effects of testosterone on muscle strength, physical function, body composition, and quality of life persist six months after treatment in intermediate-frail and frail elderly men? J. Clin. Endocrinol. Metab. 96, 454–458 (2011).
Ng Tang Fui, M. et al. Effects of testosterone treatment on body fat and lean mass in obese men on a hypocaloric diet: a randomised controlled trial. BMC Med. 14, 153 (2016).
Ohlander, S. J., Varghese, B. & Pastuszak, A. W. Erythrocytosis following testosterone therapy. Sex. Med. Rev. 6, 77–85 (2018).
Albert, S. G. & Morley, J. E. Testosterone therapy, association with age, initiation and mode of therapy with cardiovascular events: a systematic review. Clin. Endocrinol. (Oxf.) 85, 436–443 (2016).
Martinez, C. et al. Testosterone treatment and risk of venous thromboembolism: population based case-control study. BMJ 355, i5968 (2016).
Basaria, S. et al. Adverse events associated with testosterone administration. N. Engl. J. Med. 363, 109–122 (2010).
Morgentaler, A. et al. Fundamental concepts regarding testosterone deficiency and treatment: international expert consensus resolutions. Mayo Clin. Proc. 91, 881–896 (2016).
Bhasin, S. et al. Testosterone therapy in men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 95, 2536–2559 (2010).
Dubois, V., Laurent, M., Boonen, S., Vanderschueren, D. & Claessens, F. Androgens and skeletal muscle: cellular and molecular action mechanisms underlying the anabolic actions. Cell. Mol. Life Sci. 69, 1651–1667 (2012).
Crawford, J. et al. Study design and rationale for the phase 3 clinical development program of enobosarm, a selective androgen receptor modulator, for the prevention and treatment of muscle wasting in cancer patients (POWER Trials). Curr. Oncol. Rep. 18, 37 (2016).
Dobs, A. S. et al. Effects of enobosarm on muscle wasting and physical function in patients with cancer: a double-blind, randomised controlled phase 2 trial. Lancet Oncol. 14, 335–345 (2013).
Dalton, J. T. et al. The selective androgen receptor modulator GTx-024 (enobosarm) improves lean body mass and physical function in healthy elderly men and postmenopausal women: results of a double-blind, placebo-controlled phase II trial. J. Cachexia Sarcopenia Muscle 2, 153–161 (2011).
Papanicolaou, D. A. et al. A phase IIA randomized, placebo-controlled clinical trial to study the efficacy and safety of the selective androgen receptor modulator (SARM), MK-0773 in female participants with sarcopenia. J. Nutr. Health Aging 17, 533–543 (2013).
Bhattacharya, I. et al. Safety, pharmacokinetic, and pharmacodynamic evaluation after single and multiple ascending doses of a novel selective androgen receptor modulator in healthy subjects. Clin. Ther. 38, 1401–1416 (2016).
Watanabe, K. et al. BA321, a novel carborane analog that binds to androgen and estrogen receptors, acts as a new selective androgen receptor modulator of bone in male mice. Biochem. Biophys. Res. Commun. 478, 279–285 (2016).
Saeed, A. et al. 2-Chloro-4-[[(1R,2R)-2-hydroxy-2-methyl-cyclopentyl]amino]-3-methyl-benzonitrile: a transdermal selective androgen receptor modulator (SARM) for muscle atrophy. J. Med. Chem. 59, 750–755 (2016).
von Haehling, S. & Anker, S. D. Treatment of cachexia: an overview of recent developments. Int. J. Cardiol. 184, 736–742 (2015).
Garcia, J. M. & Polvino, W. J. Pharmacodynamic hormonal effects of anamorelin, a novel oral ghrelin mimetic and growth hormone secretagogue in healthy volunteers. Growth Horm. IGF Res. 19, 267–273 (2009).
Garcia, J. M. & Polvino, W. J. Effect on body weight and safety of RC-1291, a novel, orally available ghrelin mimetic and growth hormone secretagogue: results of a phase I, randomized, placebo-controlled, multiple-dose study in healthy volunteers. Oncologist 12, 594–600 (2007).
Koshinaka, K. et al. Therapeutic potential of ghrelin treatment for unloading-induced muscle atrophy in mice. Biochem. Biophys. Res. Commun. 412, 296–301 (2011).
Nishie, K., Yamamoto, S., Nagata, C., Koizumi, T. & Hanaoka, M. Anamorelin for advanced non-small-cell lung cancer with cachexia: systematic review and meta-analysis. Lung Cancer 112, 25–34 (2017).
Bai, Y. et al. Anamorelin for cancer anorexia-cachexia syndrome: a systematic review and meta-analysis. Support. Care Cancer 25, 1651–1659 (2017). This is a meta-analysis of four randomized trials demonstrating that lean body mass improved with anamorelin, a novel ghrelin receptor agonist.
Garcia, J. M. et al. Anamorelin for patients with cancer cachexia: an integrated analysis of two phase 2, randomised, placebo-controlled, double-blind trials. Lancet Oncol. 16, 108–116 (2015).
Temel, J. S. et al. Anamorelin in patients with non-small-cell lung cancer and cachexia (ROMANA 1 and ROMANA 2): results from two randomised, double-blind, phase 3 trials. Lancet Oncol. 17, 519–531 (2016).
Katakami, N. et al. Anamorelin (ONO-7643) for the treatment of patients with non-small cell lung cancer and cachexia: results from a randomized, double-blind, placebo-controlled, multicenter study of Japanese patients (ONO-7643-04). Cancer 124, 606–616 (2018).
Bergen, H. R. III et al. Myostatin as a mediator of sarcopenia versus homeostatic regulator of muscle mass: insights using a new mass spectrometry-based assay. Skelet. Muscle 5, 21 (2015).
McPherron, A. C., Lawler, A. M. & Lee, S. J. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature 387, 83–90 (1997).
White, T. A. & LeBrasseur, N. K. Myostatin and sarcopenia: opportunities and challenges — a mini-review. Gerontology 60, 289–293 (2014).
Camporez, J. P. et al. Anti-myostatin antibody increases muscle mass and strength and improves insulin sensitivity in old mice. Proc. Natl Acad. Sci. USA 113, 2212–2217 (2016). This study evaluates a treatment consisting of anti-myostatin antibody administration for 4 weeks, showing improved muscle mass and strength in young and old mice.
Dong, J. et al. Inhibition of myostatin in mice improves insulin sensitivity via irisin-mediated cross talk between muscle and adipose tissues. Int. J. Obes. (Lond.) 40, 434–442 (2016).
Buehring, B. & Binkley, N. Myostatin — the holy grail for muscle, bone, and fat? Curr. Osteoporos. Rep. 11, 407–414 (2013).
Santos, A. R. et al. Different resistance-training regimens evoked a similar increase in myostatin inhibitors expression. Int. J. Sports Med. 36, 761–768 (2015).
Sato, Y., Honda, Y., Kuno, H. & Oizumi, K. Menatetrenone ameliorates osteopenia in disuse-affected limbs of vitamin D− and K-deficient stroke patients. Bone 23, 291–296 (1998).
Schurgers, L. J. & Vermeer, C. Determination of phylloquinone and menaquinones in food. Effect of food matrix on circulating vitamin K concentrations. Haemostasis 30, 298–307 (2000).
Hart, J. P. et al. Electrochemical detection of depressed circulating levels of vitamin K1 in osteoporosis. J. Clin. Endocrinol. Metab. 60, 1268–1269 (1985).
Bitensky, L. et al. Circulating vitamin K levels in patients with fractures. J. Bone Joint Surg. Br. 70, 663–664 (1988).
Hodges, S. J., Akesson, K., Vergnaud, P., Obrant, K. & Delmas, P. D. Circulating levels of vitamins K1 and K2 decreased in elderly women with hip fracture. J. Bone Miner. Res. 8, 1241–1245 (1993).
Hodges, S. J. et al. Depressed levels of circulating menaquinones in patients with osteoporotic fractures of the spine and femoral neck. Bone 12, 387–389 (1991).
Binkley, N. et al. Vitamin K treatment reduces undercarboxylated osteocalcin but does not alter bone turnover, density, or geometry in healthy postmenopausal North American women. J. Bone Miner. Res. 24, 983–991 (2009).
Binkley, N. C. & Suttie, J. W. Vitamin K nutrition and osteoporosis. J. Nutr. 125, 1812–1821 (1995).
Caillot-Augusseau, A. et al. Space flight is associated with rapid decreases of undercarboxylated osteocalcin and increases of markers of bone resorption without changes in their circadian variation: observations in two cosmonauts. Clin. Chem. 46, 1136–1143 (2000).
Douglas, A. S. et al. Carboxylation of osteocalcin in post-menopausal osteoporotic women following vitamin K and D supplementation. Bone 17, 15–20 (1995).
Macdonald, H. M. et al. Vitamin K1 intake is associated with higher bone mineral density and reduced bone resorption in early postmenopausal Scottish women: no evidence of gene-nutrient interaction with apolipoprotein E polymorphisms. Am. J. Clin. Nutr. 87, 1513–1520 (2008).
Jamal, S. A., Browner, W. S., Bauer, D. C. & Cummings, S. R. Warfarin use and risk for osteoporosis in elderly women. Study of Osteoporotic Fractures Research Group. Ann. Intern. Med. 128, 829–832 (1998).
Yonemura, K., Kimura, M., Miyaji, T. & Hishida, A. Short-term effect of vitamin K administration on prednisolone-induced loss of bone mineral density in patients with chronic glomerulonephritis. Calcif. Tissue Int. 66, 123–128 (2000).
Yoshida, M. et al. Effect of vitamin K supplementation on insulin resistance in older men and women. Diabetes Care 31, 2092–2096 (2008).
Shea, M. K., Dawson-Hughes, B., Gundberg, C. M. & Booth, S. L. Reducing undercarboxylated osteocalcin with vitamin k supplementation does not promote lean tissue loss or fat gain over 3 years in older women and men: a randomized controlled trial. J. Bone Miner. Res. 32, 243–249 (2017).
Shah, K., Gleason, L. & Villareal, D. T. Vitamin K and bone health in older adults. J. Nutr. Gerontol. Geriatr. 33, 10–22 (2014).
Collins, C. A. et al. Stem cell function, self-renewal, and behavioral heterogeneity of cells from the adult muscle satellite cell niche. Cell 122, 289–301 (2005).
Sacco, A., Doyonnas, R., Kraft, P., Vitorovic, S. & Blau, H. M. Self-renewal and expansion of single transplanted muscle stem cells. Nature 456, 502–506 (2008).
Tompkins, B. A. et al. Allogeneic mesenchymal stem cells ameliorate aging frailty: a phase ii randomized, double-blind, placebo-controlled clinical trial. J. Gerontol. A Biol. Sci. Med. Sci. 72, 1513–1522 (2017).
Newman, A. B. et al. Sarcopenia: alternative definitions and associations with lower extremity function. J. Am. Geriatr. Soc. 51, 1602–1609 (2003). This evaluates two sarcopenia definitions in the Health, Aging and Body Composition study and suggests differential prevalence rates by obesity status, highlighting the importance of fat mass in the evaluation of sarcopenia.
Baumgartner, R. N. Body composition in healthy aging. Ann. NY Acad. Sci. 904, 437–448 (2000).
Acknowledgements
The authors would like to thank R. Masutani and R. Dokko for their help with preparing the manuscript and E. Weiss for contributing to the images in figure 2. J.A.B.’s research reported in this publication was supported in part by the US National Institute on Aging of the US National Institutes of Health under award number K23AG051681. Support was also provided by the Dartmouth Health Promotion and Disease Prevention Research Center supported by cooperative agreement number U48DP005018 from the Centers for Disease Control and Prevention and the Dartmouth Clinical and Translational Science Institute under award number UL1TR001086 from the US National Center for Advancing Translational Sciences. D.T.V.’s research was supported in part by DK109950 from the US National Institute of Diabetes, Digestive and Kidney Diseases, AG031176 from the US National Institutes on Aging, CX000906 from the Veterans Affairs Office of Research and Development, 1-14-LLY-38 from the American Diabetes Association and the Alkek Foundation. The content is solely the responsibility of the authors and does not necessarily represent the official views of the US National Institutes of Health or of the Department of Veterans Affairs, nor represents the official position of the Centers for Disease Control and Prevention.
Reviewer information
Nature Reviews Endocrinology thanks W. Kemmler and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
J.A.B. and D.T.V. both researched the data for the article, provided substantial contributions to discussion of the content, wrote the article and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Sarcopenia
-
The loss of muscle mass, strength or physical function with age.
- Oxidative capacity
-
The maximal ability of muscle to utilize oxygen per g of muscle per hour.
- Thermic effect of food
-
The amount of energy expended owing to the body’s processing and storage of food.
- Fat-free mass
-
A term interchangeably used with muscle mass and lean mass; it refers to the mass of all visceral organs, muscles (smooth and skeletal), bones, ligaments and tendons but does not include fat that is present in the marrow of bones or internal organs.
- Waist circumference
-
An anthropometric measure of central obesity (subcutaneous and visceral) measured at the level of the iliac crest.
- Visceral fat
-
A measurement of the adiposity located among organs within the abdominal cavity; it is associated with inflammation and increased cardiometabolic risk.
- Intramyocellular lipids
-
Fat depositions within the muscle structure.
- Myostatin
-
A transforming growth factor-related protein that is synthesized and secreted in skeletal muscle and negatively regulates muscle mass and function.
- VO2 max
-
The maximal amount of oxygen used per kg of body weight during maximal exercise.
- Lean mass
-
A term that refers to the mass of all visceral organs, muscles (smooth and skeletal), bones, ligaments and tendons but excludes fat from bone.
- Appendicular lean mass
-
Muscle mass consisting of the sum of the upper and lower limbs.
- Grip strength
-
A measurement used in the ascertainment of upper extremity strength; it is assessed using the dominant hand with a hand-held dynamometer.
- Knee extensor strength
-
A measure of lower extremity strength. The test is performed using a dynamometer with the participant sitting with hips and knees flexed at 90°; the participant extends his or her knee and pushes against a resistance pad — the results are measured in kilograms or pounds.
- Quadriceps muscle area
-
Cross-sectional 2D area at the level of the quadriceps muscle of the lower limb.
- Skeletal muscle index
-
Absolute muscle mass (in kg) normalized for height (muscle mass in kg divided by height (in m)).
- Absolute muscle mass
-
Muscle mass consisting of all limbs and muscle from visceral organs.
Rights and permissions
About this article
Cite this article
Batsis, J.A., Villareal, D.T. Sarcopenic obesity in older adults: aetiology, epidemiology and treatment strategies. Nat Rev Endocrinol 14, 513–537 (2018). https://doi.org/10.1038/s41574-018-0062-9
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-018-0062-9
This article is cited by
-
Reduced thoracic skeletal muscle size is associated with adverse outcomes in diabetes patients with heart failure and reduced ejection fraction: quantitative analysis of sarcopenia by using cardiac MRI
Cardiovascular Diabetology (2024)
-
Neutrophil-to-lymphocyte ratio is associated with sarcopenia risk in overweight maintenance hemodialysis patients
Scientific Reports (2024)
-
Body composition and cancer survival: a narrative review
British Journal of Cancer (2024)
-
Sarcopenic obesity in older adults: a clinical overview
Nature Reviews Endocrinology (2024)
-
Neuroprotective treatment with the nitrone compound OKN-007 mitigates age-related muscle weakness in aging mice
GeroScience (2024)