Abstract
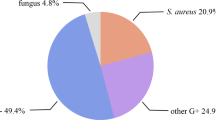
Diabetic foot ulcers are often complicated by infection. Among pathogens, Staphylococcus aureus predominates. The prevalence of methicillin-resistant S. aureus (MRSA) in infected foot ulcers is 15–30% and there is an alarming trend for increase in many countries. There are also data that recognize new strains of MRSA that are resistant to vancomycin. The risk for MRSA isolation increases in the presence of osteomyelitis, nasal carriage of MRSA, prior use of antibacterials or hospitalization, larger ulcer size and longer duration of the ulcer. The need for amputation and surgical debridement increases in patients infected with MRSA. Infections of mild or moderate severity caused by community-acquired MRSA can be treated with cotrimoxazole (trimethoprim/sulfamethoxazole), doxycycline or clindamycin when susceptibility results are available, while severe community-acquired or hospital-acquired MRSA infections should be managed with glycopeptides, linezolide or daptomycin. Dalbavancin, tigecycline and ceftobiprole are newer promising antimicrobial agents active against MRSA that may also have a role in the treatment of foot infections if more data on their efficacy and safety become available.






Similar content being viewed by others
References
King H, Aubert RE, Herman WH. Global burden of diabetes, 1995–2025: prevalence, numerical estimates, and projections. Diabetes Care 1998; 21(9): 1414–31
Boulton AJ. The diabetic foot: grand overview, epidemiology and pathogenesis. Diabetes Metab Res Rev 2008; 24 Suppl. 1: S3–6
Boulton AJ, Vileikyte L, Ragnarson-Tennvall G, et al. The global burden of diabetic foot disease. Lancet 2005; 366(9498): 1719–24
Prompers L, Huijberts M, Schaper N, et al. Resource utilisation and costs associated with the treatment of diabetic foot ulcers: prospective data from the Eurodiale Study. Diabetologia 2008; 51(10): 1826–34
Tentolouris N, Al-Sabbagh S, Walker MG, et al. Mortality in diabetic and nondiabetic patients after amputations performed from 1990 to 1995: a 5-year follow-up study. Diabetes Care 2004; 27(7): 1598–604
Marso SP, Hiatt WR. Peripheral arterial disease in patients with diabetes. J Am Coll Cardiol 2006; 47(5): 921–9
Raymakers JT, Houben AJ, van der Heyden JJ, et al. The effect of diabetes and severe ischaemia on the penetration of ceftazidime into tissues of the limb. Diabet Med 2001; 18(3): 229–34
Lipsky BA, Berendt AR, Deery HG, et al. Diagnosis and treatment of diabetic foot infections. Clin Infect Dis 2004; 39(7): 885–910
Tentolouris N, Jude EB, Smirnof I, et al. Methicillin-resistant Staphylococcus aureus: an increasing problem in a diabetic foot clinic. Diabet Med 1999; 16(9): 767–71
Lavery LA, Armstrong DG, Murdoch DP, et al. Validation of the Infectious Diseases Society of America’s diabetic foot infection classification system. Clin Infect Dis 2007; 44(4): 562–5
Cavanagh PR, Lipsky BA, Bradbury AW, et al. Treatment for diabetic foot ulcers. Lancet 2005; 366(9498): 1725–35
Chang S, Sievert DM, Hageman JC, et al. Infection with vancomycin-resistant Staphylococcus aureus containing the vanA resistance gene. N Engl J Med 2003; 348(14): 1342–7
Sievert DM, Rudrik JT, Patel JB, et al. Vancomycin-resistant Staphylococcus aureus in the United States, 2002-2006. Clin Infect Dis 2008; 46(5): 668–74
Richard JL, Sotto A, Jourdan N, et al. Risk factors and healing impact of multidrug-resistant bacteria in diabetic foot ulcers. Diabetes Metab 2008; 34 (4 Pt 1): 363–9
Ge Y, MacDonald D, Hait H, et al. Microbiological profile of infected diabetic foot ulcers. Diabet Med 2002; 19(12): 1032–4
Dang CN, Prasad YD, Boulton AJ, et al. Methicillin-resistant Staphylococcus aureus in the diabetic foot clinic: a worsening problem. Diabet Med 2003; 20(2): 159–61
Goldstein EJ, Citron DM, Nesbit CA. Diabetic foot infections: bacteriology and activity of 10 oral antimicrobial agents against bacteria isolated from consecutive cases. Diabetes Care 1996; 19(6): 638–41
El-Tahawy AT. Bacteriology of diabetic foot. Saudi Med J 2000; 21(4): 344–7
Carvalho CB, Neto RM, Aragao LP, et al. Diabetic foot infection: bacteriologic analysis of 141 patients. Arq Bras Endocrinol Metab 2004; 48(3): 398–405
Abdulrazak A, Bitar ZI, Al-Shamali AA, et al. Bacteriological study of diabetic foot infections. J Diabetes Complications 2005; 19(3): 138–41
Tentolouris N, Petrikkos G, Vallianou N, et al. Prevalence of methicillin-resistant Staphylococcus aureus in infected and uninfected diabetic foot ulcers. Clin Microbiol Infect 2006; 12(2): 186–9
Stanaway S, Johnson D, Moulik P, et al. Methicillin-resistant Staphylococcus aureus (MRSA) isolation from diabetic foot ulcers correlates with nasal MRSA carriage. Diabetes Res Clin Pract 2007; 75(1): 47–50
Lipsky BA, Stoutenburgh U. Daptomycin for treating infected diabetic foot ulcers: evidence from a randomized, controlled trial comparing daptomycin with vancomycin or semi-synthetic penicillins for complicated skin and skinstructure infections. J Antimicrob Chemother 2005; 55(2): 240–5
Candel Gonzalez FJ, Alramadan M, Matesanz M, et al. Infections in diabetic foot ulcers. Eur J Intern Med 2003; 14(5): 341–3
Citron DM, Goldstein EJ, Merriam CV, et al. Bacteriology of moderate-to-severe diabetic foot infections and in vitro activity of antimicrobial agents. J Clin Microbiol 2007; 45(9): 2819–28
Martinez-Gómez Dde A, Ramírez-Almagro C, Campillo-Soto A, et al. Diabetic foot infections: prevalence and antibiotic sensitivity of the causative microorganisms. Enferm Infecc Microbiol Clin 2009; 27(6): 317–21
Aragon-Sanchez FJ, Cabrera-Galvan JJ, Quintana-Marrero Y, et al. Outcomes of surgical treatment of diabetic foot osteomyelitis: a series of 185 patients with histopathological confirmation of bone involvement. Diabetologia 2008; 51(11): 1962–70
Shankar EM, Mohan V, Premalatha G, et al. Bacterial etiology of diabetic foot infections in South India. Eur J Intern Med 2005; 16(8): 567–70
Hartemann-Heurtier A, Robert J, Jacqueminet S, et al. Diabetic foot ulcer and multidrug-resistant organisms: risk factors and impact. Diabet Med 2004; 21(7): 710–5
Gadepalli R, Dhawan B, Sreenivas V, et al. A clinico-microbiological study of diabetic foot ulcers in an Indian tertiary care hospital. Diabetes Care 2006; 29(8): 1727–32
Raja NS. Microbiology of diabetic foot infections in a teaching hospital in Malaysia: a retrospective study of 194 cases. J Microbiol Immunol Infect 2007; 40(1): 39–44
Yates C, May K, Hale T, et al. Wound chronicity, inpatient care, and chronic kidney disease predispose to MRSA infection in diabetic foot ulcers. Diabetes Care 2009; 32(10): 1907–9
Aragon-Sanchez FJ, Lazaro-Martinez JL, Quintana-Marrero Y, et al. Are diabetic foot ulcers complicated by MRSA osteomyelitis associated with worse prognosis? Outcomes of a surgical series. Diabet Med 2009; 26(5): 552–5
Galkowska H, Podbielska A, Olszewski WL, et al. Epidemiology and prevalence of methicillin-resistant Staphylococcus aureus and Staphylococcus epidermidis in patients with diabetic foot ulcers: focus on the differences between species isolated from individuals with ischemic vs. neuropathic foot ulcers. Diabetes Res Clin Pract 2009; 84(2): 187–93
Vardakas KZ, Horianopoulou M, Falagas ME. Factors associated with treatment failure in patients with diabetic foot infections: an analysis of data from randomized controlled trials. Diabetes Res Clin Pract 2008; 80(3): 344–51
Fejfarova V, Jirkovska A, Skibova J, et al. Pathogen resistance and other risk factors in the frequency of lower limb amputations in patients with the diabetic foot syndrome. Vnitr Lek 2002; 48(4): 302–6
Nather A, Bee CS, Huak CY, et al. Epidemiology of diabetic foot problems and predictive factors for limb loss. J Diabetes Complications 2008; 22(2): 77–82
Rozgonyi F, Kocsis E, Kristof K, et al. Is MRSA more virulent than MSSA? Clin Microbiol Infect 2007; 13(9): 843–5
Mizobuchi S, Minami J, Jin F, et al. Comparison of the virulence of methicillin-resistant and methicillin-sensitive Staphylococcus aureus. Microbiol Immunol 1994; 38(8): 599–605
Wang R, Braughton KR, Kretschmer D, et al. Identification of novel cytolytic peptides as key virulence determinants for community-associated MRSA. Nature Med 2007; 13(12): 1510–4
Eady EA, Cove JH. Staphylococcal resistance revisited: community-acquired methicillin resistant Staphylococcus aureus: an emerging problem for the management of skin and soft tissue infections. Curr Opin Infect Dis 2003; 16(2): 103–24
Ho PL, Chuang SK, Choi YF, et al. Community-associated methicillin-resistant and methicillin-sensitive Staphylococcus aureus: skin and soft tissue infections in Hong Kong. Diagn Microbiol Infect Dis 2008; 61(3): 245–50
Sotto A, Lina G, Richard JL, et al. Virulence potential of Staphylococcus aureus strains isolated from diabetic foot ulcers: a new paradigm. Diabetes Care 2008; 31(12): 2318–24
Deleo FR, Otto M, Kreiswirth BN, et al. Community-associated meticillin-resistant Staphylococcus aureus. Lancet 2010; 375(9725): 1557–68
Lee MC, Rios AM, Aten MF, et al. Management and outcome of children with skin and soft tissue abscesses caused by community-acquired methicillin-resistant Staphylococcus aureus. Pediatr Infect Dis J 2004; 23(2): 123–7
Jude EB, Unsworth PF. Optimal treatment of infected diabetic foot ulcers. Drugs Aging 2004; 21(13): 833–50
Bowling FL, Salgami EV, Boulton AJ. Larval therapy: a novel treatment in eliminating methicillin-resistant Staphylococcus aureus from diabetic foot ulcers. Diabetes Care 2007; 30(2): 370–1
Dang CN, Anwar R, Thomas G, et al. The Biogun: a novel way of eradicating methicillin-resistant Staphylococcus aureus colonization in diabetic foot ulcers [letter]. Diabetes Care 2006; 29(5): 1176
Lipsky BA. The Biogun: a novel way of eradicating methicillin-resistant Staphylococcus aureus colonization in diabetic foot ulcers. Response to Dang et al. [letter]. Diabetes Care 2006; 29(9): 2181–2
Niu SC, Deng ST, Lee MH, et al. Modified vancomycin dosing protocol for treatment of diabetic foot infections. Am J Health Syst Pharm 2008; 65(18): 1740–3
Adam HJ, Louie L, Watt C, et al. Detection and characterization of heterogeneous vancomycin-intermediate Staphylococcus aureus isolates in Canada: results from the Canadian Nosocomial Infection Surveillance Program, 1995–2006. Antimicrob Agents Chemother 2010; 54(2): 945–9
Sader HS, Fey PD, Limaye AP, et al. Evaluation of vancomycin and daptomycin potency trends (MIC creep) against methicillin-resistant Staphylococcus aureus isolates collected in nine U.S. medical centers from 2002 to 2006. Antimicrob Agents Chemother 2009; 53(10): 4127–32
Rybak MJ, Lerner SA, Levine DP, et al. Teicoplanin pharmacokinetics in intravenous drug abusers being treated for bacterial endocarditis. Antimicrob Agents Chemother 1991; 35(4): 696–700
Edmonds M. The treatment of diabetic foot infections: focus on ertapenem. Vascular Health Risk Manag 2009; 5: 949–63
Fitch L, Johnson AP. Reduced susceptibility to teicoplanin in a methicillin-resistant strain of Staphylococcus aureus [letter]. J Antimicrob Chemother 1998; 41(5): 578
Stein GE, Schooley S, Peloquin CA, et al. Linezolid tissue penetration and serum activity against strains of methicillin-resistant Staphylococcus aureus with reduced vancomycin susceptibility in diabetic patients with foot infections. J Antimicrob Chemother 2007; 60(4): 819–23
Omar NS, El-Nahas MR, Gray J. Novel antibiotics for the management of diabetic foot infections. Int J Antimicrob Agents 2008; 31(5): 411–9
Majcher-Peszynska J, Haase G, Sass M, et al. Pharmacokinetics and penetration of linezolid into inflamed soft tissue in diabetic foot infections. Eur J Clin Pharmacol 2008; 64(11): 1093–100
Nelson SB. Management of diabetic foot infections in an era of increasing microbial resistance. Curr Infect Dis Rep 2009; 11(5): 375–82
Goldstein EJ, Citron DM, Warren YA, et al. In vitro activities of dalbavancin and 12 other agents against 329 aerobic and anaerobic gram-positive isolates recovered from diabetic foot infections. Antimicrob Agents Chemother 2006; 50(8): 2875–9
Sotto A, Bouziges N, Jourdan N, et al. In vitro activity of tigecycline against strains isolated from diabetic foot ulcers [in French]. Pathol Biol (Paris) 2007; 55(8-9): 398–406
Goldstein EJ, Citron DM, Merriam CV, et al. In vitro activity of ceftobiprole against aerobic and anaerobic strains isolated from diabetic foot infections. Antimicrob Agents Chemother 2006; 50(11): 3959–62
Wang J, Soisson SM, Young K, et al. Platensimycin is a selective FabF inhibitor with potent antibiotic properties. Nature 2006; 441(7091): 358–61
Steed ME, Rybak MJ. Ceftaroline: a new cephalosporin with activity against resistant gram-positive pathogens. Pharmacotherapy 2010; 30(4): 375–89
Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft-tissue infections. Clin Infect Dis 2005; 41(10): 1373–406
Lewis 2nd JS, Jorgensen JH. Inducible clindamycin resistance in Staphylococci: should clinicians and microbiologists be concerned? Clin Infect Dis 2005; 40(2): 280–5
Naimi TS, LeDell KH, Como-Sabetti K, et al. Comparison of community- and health care-associated methicillin-resistant Staphylococcus aureus infection. JAMA 2003; 290(22): 2976–84
Lipsky BA. Empirical therapy for diabetic foot infections: are there clinical clues to guide antibiotic selection? Clin Microbiol Infect 2007; 13(4): 351–3
Lipsky BA. A report from the international consensus on diagnosing and treating the infected diabetic foot. Diabetes Metab Res Rev 2004; 20 Suppl. 1: S68–77
D’Agostino Sr RB, Massaro JM, Sullivan LM. Non-inferiority trials: design concepts and issues-the encounters of academic consultants in statistics. Stat Med 2003; 22(2): 169–86
Snapinn SM. Noninferiority trials. Curr Control Trials Cardiovasc Med 2000; 1(1): 19–21
Lecornet E, Robert J, Jacqueminet S, et al. Preemptive isolation to prevent methicillin-resistant Staphylococcus aureus cross-transmission in diabetic foot. Diabetes Care 2007; 30(9): 2341–2
Wagner A, Reike H, Angelkort B. Highly resistant pathogens in patients with diabetic foot syndrome with special reference to methicillin-resistant Staphylococcus aureus infections. Dtsch Med Wochenschr 2001; 126(48): 1353–6
Lagace-Wiens PR, Ormiston D, Nicolle LE, et al. The diabetic foot clinic: not a significant source for acquisition of methicillin-resistant Staphylococcus aureus. Am J Infect Control 2009; 37(7): 587–9
Sotto A, Richard JL, Jourdan N, et al. Miniaturized oligonucleotide arrays: a new tool for discriminating colonization from infection due to Staphylococcus aureus in diabetic foot ulcers. Diabetes Care 2007; 30(8): 2051–6
Pellizzer G, Strazzabosco M, Presi S, et al. Deep tissue biopsy vs. superficial swab culture monitoring in the microbiological assessment of limb-threatening diabetic foot infection. Diabet Med 2001; 18(10): 822–7
Acknowledgements
No sources of funding were used to assist in the preparation of this review. The authors have no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eleftheriadou, I., Tentolouris, N., Argiana, V. et al. Methicillin-Resistant Staphylococcus aureus in Diabetic Foot Infections. Drugs 70, 1785–1797 (2010). https://doi.org/10.2165/11538070-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11538070-000000000-00000